INTRODUCTION
Principal mood disorders i.e., major depressive disorder (MDD) and bipolar disorder (BD) are severe neuropsychiatric ailments exemplified, in most cases, by onset in adolescence or early adulthood, recurrent course and a life-long duration. Due to persistent fluctuations, the patients experience constant variations in energy levels, affect, sleep and the psychological status [1]. This diathesis has an inclusive negative influence on all aspects of lives, as patients experience difficulties in social, psychological, academic and occupational realms of functioning. Accumulating evidence indicates that mood disorders are progressive conditions, so that there is a great need to stage define affective illnesses, since this is helpful in delivering tailored treatments to the patients [2].
In many cases the trajectory of mood disorders is typified by epochs, such as childhood precursor disorders, a clinical symptomatic state and a residual stage of functional deficits. Researchers have underscored the difficulty in the initial identification and, the resulting delay in the accurate diagnosis of mood disorders, which is of the magnitude of several years in many patients. This results in inadequate or inappropriate treatment, and a consequent exacerbation of the diathesis [3]. Therefore, patients suffer repeated mood flare-ups, inter-episode sub-threshold symptoms, and the development of such complications as substance abuse. This sequence of disease advancement leads to cognitive impairment, functional decline and loss of essential skills required in the performance of daily tasks [4]. There is growing evidence that manic and major depressive episodes have a toxic effect on brain elements such as the neurons and the glia, and produce enduring pathological changes in regions critical to mood regulation. The progressive nature of affective disorders can be arrested with early diagnosis, proper treatment on the biopsychosocial lines, recognition and management of neuropsychiatric and physical comorbidities and personalized care according to the phase of the illness [5].
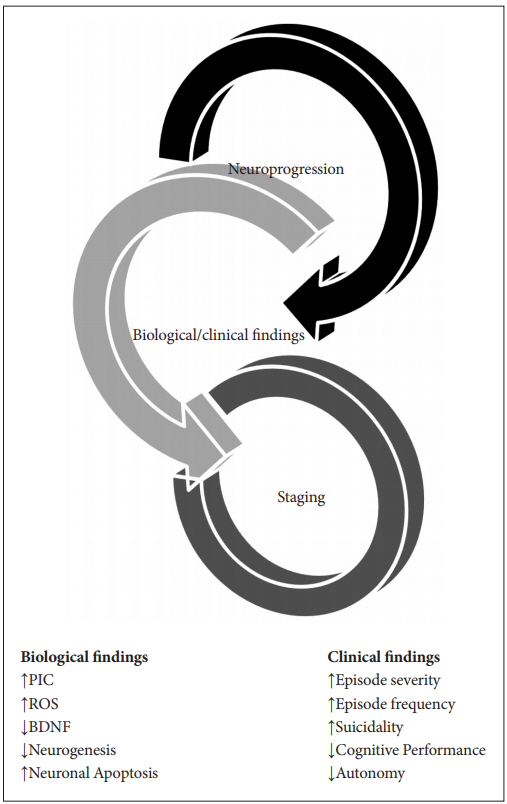
Accordingly, it is imperative to attempt to demarcate the stages of MDD and BD to facilitate the delivery of improved patient care and attain better outcomes. It must be emphasized that the staging approach has been very beneficial in the overall management of other complex (gene×environment) disorders including diabetes, cardiovascular diseases and various forms of cancer. Staging assumes that illness advances in a certain sequential progression of phases from an at-risk, prodromal period to the first clinical manifestations, and further developing into more severe, intractable forms with evolution and chronicity [6]. Figure 1 depicts the basic notions underlying the staging paradigm and shows that mood disorders have a phased evolution, ultimately leading to irreversible alterations in the brain and periphery with resulting deficits in the biopsychosocial spheres of functioning (Figure 1).
Current nosological classifications in psychiatry emphasize the presenting symptoms and have a categorical and crosssectional design. They do not take into consideration the context, the preceding risk factors, and the natural course of illness development. More than a century ago, Kraepelin first recognized that psychiatric disorders had a staged development and his view of their progressive nature was reflected in his nomenclature of dementia praecox and manic depressive insanity. Regarding the latter, he realized that while the initial episodes were frequently precipitated by stressful events followed by complete recovery, as the illness advanced patients had affective recurrences even in the absence of stressful experiences with continued psychosocial impairments [7].
Clinical staging is a valuable conceptual framework to refine the phenotype of major mood disorders along developmental lines, and supports the identification of trait and state specific biomarkers. It paves the way for stratified and individualized treatment at each stage of the illness as is already occurring in other branches of medicine. With regards to progressive conditions like mood disorders, early treatment is likely to be simpler and more successful. Furthermore, early interventions attempt to prevent or significantly delay advancement to more severe presentations which necessitate complex management strategies likely to be less acceptable to the patients, and have a lower benefit-to-risk ratio [8]. In spite of the evident benefits of clinical staging, this facet of illness classification has been largely neglected in psychiatry until recently. Nevertheless, now there is an increased interest to characterize by stages major mood disorders [9]. In this respect, staging models have been proposed by different researchers incorporating the longitudinal history of affective illnesses from a high, at-risk period starting in childhood to illness progression to more severe and refractory states in later life. Here, a sincere effort is made to portray the latest understanding of the staging models as this applies to mood disorders, and the associated neurobiological alterations are presented. The clinical and biological findings are highlighted to reconcile the two phenomena, and the practical advantages of incorporating such notions in every day practice are emphasized.
SEARCH STRATEGY
In October 2017, the PUBMED electronic data base was scrutinized to explore the extant literature on the subject of staging of mood disorders. A combination of various search terms was used to comprehensively explore the database. Some examples included “mood disorders and staging,” “mood disorders and neuroprogression,” “mood disorders and allostatic load,” “bipolar disorder and staging,” and “major depressive disorder and staging.” More than three hundred articles were retrieved which included papers in English and other languages. Only the former were included in subsequent considerations and importance was given to original papers and review articles. Papers which were considered to be particularly pertinent were read in full and their reference lists were also consulted. In order to incorporate the latest notions search was limited to the last 10 years, and in this manner a representation emerged which was both current and evidence based. Finally, in April 2018 a further analysis of the PUBMED database was undertaken during the process of revision of the manuscript to include the literature published in the meantime. Thus, a wide-ranging appraisal of the existing literature was performed and the findings were put together in a succinct and concise style. However, it must be stated that the methodology recommended for the writing of systematic reviews, for instance Preferred Reporting of Items for Systematic Reviews and Meta-analyses (PRISMA statement) was not followed and this is a limitation of the present paper.
DEVELOPMENTAL TRAJECTORY OF MOOD DISORDERS
Classical, recurrent bipolar disorder
Currently viewed as a spectrum illness, BD is considered to include several phenotypes. The classic BD is characterized by recurrent mood episodes which could be of either manic or depressive polarity, full inter-episode recovery, very high heritability, and good response to lithium in up to 80% of the cases [10]. It is not associated with cognitive and functional decline, and there is a relative absence of psychiatric comorbidities like anxiety and substance use disorders. The family history of patients with classical BD is discernible by high genetic loading in that they usually have a first degree relative with a related condition. As children and adolescents they may show an excess of adjustment, sleep and anxiety disorders such as circadian rhythm sleep disorders and separation anxiety disorder [11]. The offspring of classical, lithium-responsive bipolar patients are often gifted, have a completely normal early development with good academic and social adjustment. In rare cases classic, lithium-responsive BD may occur in individuals without a family history of the condition but this would be an exception rather than the rule. The frequency of recurrences and the quality of remission are known to vary substantially between patients; however, over time there is no significant change in illness phase and polarity, absence of cycle acceleration and lack of sub-threshold affective symptoms between episodes. This phenotype is marked by an absence of evidence for progressive worsening of the course; furthermore, in the family history there may be an increased incidence of recurrent mood disorders, but not schizophrenia spectrum conditions [12]. To provide an estimate, in an outpatient investigation in a Canadian teaching hospital subspecialty clinic the sample of classical BD patients comprised of 30% of the total bipolar cases studied [13]. The conventional, episodic, lithium responsive subtype now forms part of a much broader spectrum of BD.
Bipolar spectrum disorders
Evidence supports that bipolar subtypes discerned by either prophylactic response or non-response to lithium differ in clinical course, family history and neurobiological correlates. Lithium nonresponsive conditions are chronic disorders that better respond to atypical neuroleptics, and have similarities to psychotic spectrum disorders [14]. Contrarily, some bipolar spectrum patients preferentially respond to anticonvulsants, as is the case with BD type II patients with comorbid anxiety disorders [15]. BD with psychotic symptoms has overlapping neurobiological findings with schizophrenia spectrum disorders, and such patients present with a higher risk of having family members with chronic, non-affective psychoses [16]. In help seeking symptomatic youth, the illness trajectory of non-classical BD is marked by negative symptoms like affective flattening, anhedonia, apathy and social withdrawal, merging clinically with psychotic disorders. These cases may show neurodevelopmental disorders including learning disabilities, attentional problems, and academic failure and often demonstrate cluster A personality traits [17]. The illness course of such youth may be progressive, marked by mixed episodes, incomplete remissions, unrelenting subthreshold symptoms, cognitive impairments and functional decline in the latter stages of the illness. There is evidence for a persistent pro-inflammatory state, increased level of oxidative stress and failing neurotrophic support [18]. Neuroimaging studies show increased volume of lateral ventricles with loss of brain tissue in the final stages of the disease [19].
Youth forerunners of bipolar disorder
The diagnosis of BD is not established until the occurrence of the first manic or hypomanic episode; however, the bipolar diathesis most often starts with a depressive episode which may precede the first diagnosable activated episode by many years. A positive family history informs the risk of BD in youth who present with melancholic depressive episodes [20]. There are other antecedents, which comprise non-specific anxiety syndromes and persistent mood instability recognized only as depressive disorder not otherwise specified, cyclothymia, or emotional dysregulation. In addition, in a sub-set of help seeking youth attenuated psychotic symptoms, attentional and cognitive problems often co-exist along with affective symptoms [21]. The early precursors are vague and seen in other illness trajectories including psychotic disorders, internalizing and externalizing disorders and substance use disorders. These can also be self-limited presentations in some young people and may not advance to major psychiatric illnesses [22]. However, it is the nature of the family history that brings the required perspective to the otherwise indistinct clinical manifestations in children and adolescents. The history of BD in close family members and their response to specific treatments, for example, lithium are important predictive factors aiding in the early differential diagnosis and the provision of appropriate treatment to symptomatic youth [23]. In the instance, stimulants and antidepressants should be either avoided or if used, kept to low doses, short duration and be closely monitored in young patients with an established family history of BD.
Longitudinal studies
Importantly, a number of prospective studies of offspring of parents with confirmed diagnosis of BD have shown variance in illness course based on subtypes of the illness. Specifically, children of BD parents with an excellent response to long-term lithium had a completely normal or endowed early developmental course. In contrast, the children of parents who failed to respond to lithium prophylaxis manifested childhood problems with cognition, emotional regulation, socialization, and had neurodevelopmental disorders [24]. Among highrisk offspring who developed major mood disorders, the children of lithium responders had an episodic, remitting course and response to lithium prophylaxis, while the offspring of lithium non-responders showed a persistent course and a somewhat better therapeutic outcome to anticonvulsant mood stabilizers and atypical antipsychotics [25]. Additionally, the spectrum of end-stage disorders among the afflicted offspring of lithium responders was restricted to mood disorders, while the offspring of lithium non-responders manifested both mood and psychotic spectrum disorders. This analysis, based on up to 17 years of prospective clinical research observations, demonstrates that the bipolar diathesis advances in a predictable fashion. Nonetheless, it must be appreciated that not all offspring follow the same trajectory of disease development, although after entering the model the majority progress in an evident manner [26]. As the staging model is an overall clinical construct, it should be clear that not all highrisk offspring pursue an identical illness course, nor does everyone advance to end-stage illness.
Major depressive disorder
By comparison, MDD or unipolar depression is a somewhat more homogeneous condition. The age of onset is usually latter than BD, in the twenties and early thirties and the patients follow a relapsing and remitting course, exhibiting only major depressive episodes. The prevalence of general medical and neuropsychiatric comorbidities is also lower [27]. There is a high rate of conversion to BD type II or BD type I on longitudinal follow up, and even in patients who continue to have unipolar depressive episodes, many show additional benefit with the adjunctive use of mood stabilizers and atypical antipsychotics along with standard antidepressants [28]. There is definite evidence that severe, recurrent major depressive episodes lead to neuroprogressive changes in the brain, and in this regard atrophy of the hippocampal neurons and decreased neurogenesis in the dentate gyrus are well replicated findings [29]. In view of the extant evidence it is fair to assume that MDD is also an advancing diathesis amenable to the staging paradigm, with essentially similar connotations for the two principal mood disorders.
NEUROPRESSION-THE BASIS OF STAGING MOOD DISORDERS
There is increasing proof that affective illnesses are evolving conditions in which early stages have different clinical features compared to latter phases. The advancement of the diathesis frequently connected with such factors as misdiagnosis and inadequate treatment, is associated with greater frequency and severity of episodes. Furthermore, there is evidence that this phenomenon also occurs spontaneously with disease progression [30]. Recurrent and severe affective episodes are linked to higher rates of comorbidities, increased risk of suicide, more hospitalizations and overall treatment resistance. Connected to these findings are cognitive deterioration and functional decline in such areas as occupation, social relationships, and financial independence with ultimate loss of self-sufficiency [31]. Research efforts in the last few decades have begun to unravel the neurobiological correlates of the worsening clinical course in mood disorders. The term “neuroprogression” has been increasingly used to define the pathological reorganization of the central nervous system which results from such mechanisms as inflammation and oxidative stress. In affective disorders, neural substrate reactivity is changed by repeated mood episodes mediated by biological alterations. This ultimately promotes brain restructuring leading to an augmented susceptibility to life stresses [32]. In afflicted individuals an episode-dependent deterioration pattern has been extensively reported in serum biomarkers, brain imaging and neuropsychological performance [33]. Staging which attempts to demarcate the illness course into phases can be regarded as another facet of neuroprogression [34]. Figure 2 is a schematic representation of this conceptual framework and links neurobiological findings to clinical manifestations in advancing phases of affective ailments (Figure 2).
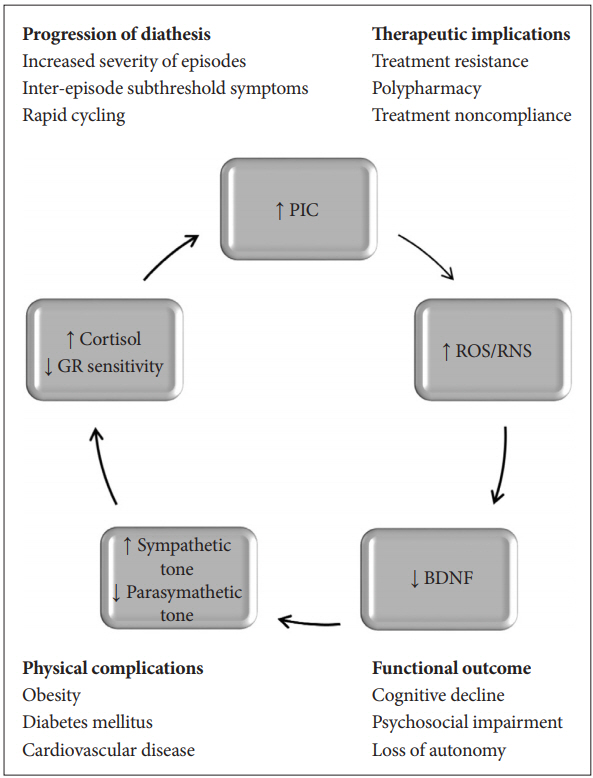
ALLOSTATIC LOAD MODEL OF MOOD DISORDERS
Mood disorders are complex and multifaceted diseases with genetic, epigenetic and environmental factors contributing to illness expression. These can also be regarded as diatheses involving the cumulative build-up of allostatic states, where physiological ‘wear and tear’ described as “allostatic load” (AL) progressively increases as stressors and mood episodes occur over time [35]. Affective episodes acting as allostatic states generate disease burden that is responsible for illness progression, as is generally seen in major mood disorders. AL may contribute to a better understanding of the disease process, particularly to vulnerability to substance abuse, decline in psychosocial functioning, cognitive dysfunction and affliction with general medical comorbidities [36]. There are pathological shifts in key mediators of AL, and include such irregularities as deregulation of the hypothalamic-pituitary-adrenal (HPA) axis, dysfunctional immune response with a continual pro-inflammatory state, increased oxidative stress and decreased neuronal survival due to the withdrawal of neurotrophic support [37]. Worse during the acute phases of the disease, these deleterious changes remain sub-threshold even during remission and damage the CNS, since the neurons are very susceptible to the mediators of AL. Whereas, the body tries to preserve homeostatic balance in the existence of escalating stress, rising AL overwhelms the compensatory physiological mechanisms, resulting in lasting damage to the brain and peripheral organs [38]. Figure 3 is a schematic representation of the pernicious nature of the advancement process unleashed by the mediators of allostatic load, which cause incremental damage to all systems of the body (Figure 3).
NEUROBIOLOGICAL FINDINGS
Neuroimaging findings
Many studies have reported the existence of changes in the brain structures of patients with major mood disorders. In BD and MDD morphometric studies found enlargements of lateral and 3rd ventricles which were not evident from the onset of the disease and were only found after several mood episodes [39]. A study in BD patients reported that the volume of the ventricles during the first manic episode was similar to that of healthy individuals, but lateral ventricles were 122% larger 8 years after disease onset in cases with multiple affective episodes [40]. Other studies have found a progressive diminution in hippocampal volume in multiple episode patients with MDD. As hippocampal volume was normal at disease outset and decreased only after repeated episodes, this may be a state marker of MDD [41].
In principal mood disorders alterations in distinctive regions of gray matter were described in a recent meta-analysis. The commonly reduced gray matter regions in both MDD and BD included bilateral insula, right superior temporal gyrus, bilateral anterior cingulate cortex, and left superior medial frontal cortex. Relatively, the volumes of right cerebellar vermis, right middle frontal gyrus, left inferior parietal lobule, and left hippocampus were significantly decreased in MDD compared to BD. Regarding white matter regions, the genu of the corpus callosum, which extended to the left prefrontal white matter, was decreased in both MDD and BD compared to healthy controls [42]. BD patients had significantly decreased fractional anisotropy values in the posterior cingulum compared to MDD patients. The primarily decreased gray matter in MDD and white matter in BD implied discrepant brain structure roles in the two major mood disorders. Cortical thickness as measured by voxel-based morphometry represents the shortest distance between the boundary of gray/white matter and the pial surface, while gray matter volume signifies the permutation of two genetically distinct anatomical attributes i.e., cortical surface area and thickness. A recent metaanalysis showed that the rostral middle frontal cortex was thinned in BD compared to MDD [43]. The often thinned cortical regions among MDD and BD cases included the left inferior temporal cortex and left fusiform gyrus.
Finally, many studies have pointed to the presence of white matter hyperintensities in individuals with BD. The incidence of these abnormalities in deep white matter is about 2.5 times commoner in BD patients than in healthy controls. One study observed that the number of total brain hyperintensities increased with the number of manic episodes. A well cited meta-analysis suggested that these lesions should be used as trait markers because white matter hyperintensities were frequently seen in family members who did not have BD [44]. As a concluding note, it must be mentioned that machine learning algorithms are being developed in earnest, with structural MRI as the basis, to help in the differential diagnosis and management of major mood disorders.
Brain derived neurotrophic factor (BDNF)
Neurotrophic factors are vital for neuronal differentiation and survival and play a key role in dendritic growth, synaptogenesis, and plasticity processes like long-term potentiation. BDNF is highly concentrated in limbic and paralimbic areas; it is understood that serum BDNF levels compare with brain BDNF concentrations, so that peripheral fluctuations are a sensitive marker of central disease activity. A recent metaanalysis specified that BD patients in the manic or depressive phase had lower blood concentrations of BDNF than healthy individuals, and that serum BDNF levels in euthymic patients were not notably different from controls [45]. Reduction in serum BDNF level may be a likely biomarker of acute episodes and could distinguish patients in a manic or depressive state from those in a euthymic phase at a sensitivity of 90% and a specificity of 85% [46]. Another study observed that laboratory measurements of serum BDNF concentrations may discriminate bipolar from unipolar depression at a diagnostic accuracy of 88% for values equal to or lower than 0.26 pg/mL. The authors compared plasma BDNF concentrations of BD patients in a depressive state with unipolar depression cases, and found that the first group showed lower levels of BDNF [47]. Since clinically BD can only be diagnosed with a history of a manic or hypomanic episode, the differentiation between bipolar and unipolar depression is often difficult, and serum BDNF concentrations may be an important marker in the differential diagnosis of the two conditions. Other studies proposed that BDNF may be a biomarker of disease progression and severity. In this regard, BDNF values were inversely correlated with episode severity in BD. To investigate the potential of serum BDNF in discriminating advanced and initial BD states, researchers showed that serum BDNF concentrations may differentiate patients with BD for less than 3 years from patients that had the disease for over 10 years, at a sensitivity of 100%, specificity of 88% and accuracy of 95% for values equal to or lower than 0.62 pg/mL [48]. Another potential use of serum BDNF levels is the prediction of response to treatment and the evaluation of the efficacy of psychopharmacological agents. In this regard, studies have found that there is an increase and normalization of peripheral BDNF levels after appropriate treatment of acute mood episodes. Therefore, it is fair to conclude that serum BDNF is a promising marker of mood disorders and further research is likely to validate its use in clinical practice.
Oxidative stress
Oxidative stress can be defined as the imbalance between pro and antioxidants which results in the generation of increased quantities of free radicals, such as reactive oxygen and nitrogen species (ROS & RNS). These cause cellular damage by reacting with macromolecules such as lipids, proteins, carbohydrates and DNA. The CNS is particularly susceptible to oxidative injury, because the brain uses a great amount of oxygen promoting the formation of ROS, and additionally has a limited antioxidant capacity [49].
Glutathione is the main antioxidant in the brain, while the enzymes composing the antioxidant system are most importantly, superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase. The role of oxidative stress in the pathophysiology of mood disorders has been investigated in several studies, which consistently report changes in antioxidant enzymes, lipid peroxidation and free radical damage to the DNA. In this vein, a meta-analysis concluded that oxidative stress markers were elevated in plasma or blood cells of individuals with BD, particularly nitric oxide and thiobarbituric acid reactive substances (TBARS) [50]. The analysis of blood TBARS (markers of lipid peroxidation) concentrations in patients with BD revealed that these were increased during all stages of the disease. This suggested that lipid damage occurred all along the course of BD and therefore, serum levels of TBARS could be used as biomarkers of progression, with the caveat that these showed increase with illness duration [51]. In addition, concentrations of nitric oxide were also elevated in BD regardless of the mood state, and that levels of 3-nitrotyrosine, another oxidative stress marker, were raised even in the early stages of the disorder [52].
One of the consequences of oxidative stress is DNA damage, and studies in patients with affective disorders have shown an increase in this aberration. In bipolar disorder, a study described a positive correlation between recurrent manic episodes and enhanced DNA damage [53]. In this vein, DNA damage has also been associated with reductions in telomere length in lymphocytes of individuals with mood disorders which is indicative of cumulative oxidative stress. Moreover, a study in patients with BD concluded that the number of shortened telomeres and mean telomere length were associated with the number of depressive episodes along life, and interestingly association with anxiety spectrum disorders contributed to this risk [54]. Physiologically, telomere shortening occurs after each cell division as part of normal ageing. However, telomere shortening found in individuals with mood disorders is suggestive of ageing acceleration of the magnitude of 10 years. This finding may elucidate the relationship between affective illnesses and early onset of medical comorbidities in mood disorder patients.
An additional factor to support the role of oxidative stress in BD and MDD is the fact that antioxidant compounds, such as N-acetylcysteine (NAC) are advantageous in the treatment of these conditions. NAC, a glutathione precursor efficiently reduces depressive symptoms in patients with BD and improves their functioning and quality of life [55]. Furthermore, treatment effects of mood stabilizers seem to be associated with their action upon oxidative stress pathways. Patients with BD treated with lithium have a significant reduction of TBARS concentrations and of the SOD/CAT ratio, whereas healthy individuals exposed to lithium do not show the same changes. This may be secondary to lithium’s inhibition of an important enzyme of the human kinome, namely GSK3β with consequent antioxidant effects favorable for the treatment of BD [56].
Lastly, to evaluate systemic toxicity in mood disorders a study compared the concentrations of several markers in patients with BD, healthy individuals and patients with sepsis. One of their conspicuous findings was the striking scale of oxidative damage during acute episodes, which in some cases was as high as the level seen in patients with sepsis [57]. In view of the importance of peripheral oxidative stress markers, there is a need for further studies so that these could be validated for use in day to day practice.
Inflammatory factors
Inflammatory mediators i.e., cytokines are proteins or glycoproteins secreted by cells of the immune system in response to noxious stimuli. Cytokines gain access to the CNS via the periphery, while microglia the resident brain macrophages also secrete these mediators locally. Pro-inflammatory cytokines (PIC) like tumor necrosis factor alpha (TNF-α) bind to neuronal death receptors and recruit Fas (first apoptosis signal) to activate caspase 8, a pro-apoptotic enzyme leading to cell death [58]. In mood disorder patients with repeated episodes, the increased neuronal loss via apoptosis has been documented as decrease in hippocampal volume and reduction in prefrontal gray matter. In this regard, recent studies have established the existence of a chronic inflammatory state in both MDD and BD. In general, affective episodes may be considered as pro-inflammatory conditions; for example, one study revealed that patients with a manic exacerbation had higher interleukin-2 (IL-2), IL-4 and IL-6 concentrations than controls, whereas those suffering from bipolar depression showed elevated levels of IL-6 only. During euthymic periods merely IL-4 levels were raised, suggesting that mania and to lesser degree depression was associated with a proinflammatory state [59]. Another study observed that patients in a manic state had significantly higher concentrations of IL-6, TNF-α and hs-CRP than individuals without BD. After 6 weeks of treatment with mood stabilizing drugs, the same patients had decreased levels of the said inflammatory markers. However, hs-CRP values correlated significantly with reduction in manic symptomatology, suggesting that this was the most dependable pointer of treatment response [60].
To extend this argument further, preliminary studies have found that inflammatory biomarkers are capable of discriminating mood disorder patients in the initial phases of the disease from those in advanced stages. According to one study at advanced BD stages serum IL-10 concentrations (an anti-inflammatory cytokine) declined and plasma TNF-α levels increased significantly [61]. Another study found that when TNF-α concentrations were equal to or greater than 20.36 pg/mL, patients with BD at advanced stages could be discriminated from patients in initial stages at an accuracy of 91% and sensitivity and specificity of 97% and 85% respectively, indicating that TNF-α might be a potential biomarker of BD progression [62]. Increased pro-inflammatory cytokines are correlated with high rates of medical comorbidities, particularly cardiovascular diseases as inflammatory abnormalities are risk factors for the development of these conditions, as well as the metabolic syndrome [63]. Furthermore, medical burden is associated with all phases of mood disorders, exemplified by the fact that over 70% of young afflicted individuals are estimated to need treatment for chronic medical conditions. In contrast, middle-aged patients develop heart disease and atherosclerosis at a younger age than adults without mood disorders, emphasizing that general medical comorbidity is an essential component of the diathesis and should be included in the staging process [64].
STAGING MODELS
Model for BD proposed by Berk et al. [65]
a) BD initiates with an asymptomatic phase (stage 0), which is directly connected to specific risk factors, for example family history of the illness and substance abuse.
b) Stage 1 is divided into 1a and 1b, characterized by the presence of mild, nonspecific mood symptoms, with the possibility of evolving to prodromal patterns.
c) Stage 2 is where the 1st episode takes place, in either the manic or depressive phase - the latter being more common.
d) Stage 3, divided into three phases, is characterized by the first relapse or the persistence of subclinical symptoms (phase 3a), an illness course with borderline symptoms (3b), or a remitting and recurring pattern (3c).
e) Finally, stage 4 refers to patients who do not achieve full remission of symptoms, or are refractory to treatment.
Staging model of BD by Kapczinski et al. [66]
This model shifts the emphasis to functioning in the interepisode as a means to stage defining BD, along with the characterization of biomarkers for each phase.
a) The patients are classified into 5 stages, starting with a latent phase in which individuals may experience mood and anxiety symptoms without the overt presence of BD.
b) In stage 1, patients have already suffered an episode of mania or hypomania, but maintain adequate functioning with absence of cognitive deficits in the inter-episode.
c) In stage 2, subclinical symptoms, comorbidities and mild cognitive dysfunction are present in the inter-episode.
d) In stage 3, patients have marked cognitive and functional impairments but retain self-independence.
e) Finally, stage 4 is characterized by marked cognitive and functional deterioration, leading to loss of autonomy for the patients.
Duffy’s Model of BD [67]
Recently, Duffy came up with her version of staging, and according to this format patients would first be classified into either classical or bipolar spectrum disorders. Each group could then have the following possible stages:
a) Stage 0 or prodromal phase where susceptible individuals are well, but with a positive family history in 1st degree relatives.
b) Stage 1 occurring in children or adolescents is marked by anxiety, sleep and adjustment disorders which are of severe nature in bipolar spectrum cases as compared to classical, lithium responsive patients.
c) Stage 2, having onset in young adulthood is characterized by circumscribed mood episodes, primarily of a depressive nature. However, in bipolar spectrum patients with history of psychosis in 1st degree relatives there could be deficit symptoms like amotivation, lack of affect and anhedonia.
d) Stage 3 is characterized by remitting manic and depressive episodes responsive to lithium in classical BD. By contrast, bipolar spectrum patients experience psychotic symptoms, cognitive deficits and psychosocial decline.
e) Stage 4 is marked by euphoric mania in classical BD, whereas there are mixed or psychotic features in bipolar spectrum patients.
f) The final stage in classical BD is characterized by some neuropsychiatric and medical comorbidities but the illness still remains responsive to lithium and has an overall better prognosis. In contradistinction, the bipolar spectrum group shows features reminiscent of schizophrenia spectrum disorders with positive psychotic symptoms, cognitive decline and functional deterioration.
Stage defining MDD
Recently Ferensztajn et al. [68] put forward a staging proposal for MDD with its basis on the notions of allostatic load and neuroprogression.
a) Stage 1 is characterized by the preclinical phase where there are such risk factors as positive family history of MDD in 1st degree relatives (1a), or there are non-specific neuropsychiatric symptoms not achieving the level of major depressive episode (1b).
b) In stage 2 there is the occurrence of a full-blown major depressive episode but with complete remission of symptoms.
c) Stage 3 is characterized by ill-defined residual symptoms (3a), or dysthymia (3b) in the inter-episode.
d) Stage 4 is marked by recurrent major depressive episodes (4a), or “double depression” if dysthymia is present (4b). In other words, there is the persistence of sub-threshold depressive symptoms between episodes.
e) In stage 5 there is the presence of “chronic depression,” meaning that the duration of depressive episode is for 2 years or longer.
TREATMENT IMPLICATIONS OF STAGING
In other branches of medicine staging is extensively employed to implement appropriate treatment strategies and inform the prognosis. However, this is an emerging trend in psychiatry and only recently have the first models of staging been developed. For such designs to have heuristic value, the course of the disease should be predictable along time, and the illness at the initial stages must be associated with simpler treatment requirements and a better outcome. In addition, treatment efficacy should be greater at the beginning of the disease and a subsequent progressive decline should occur, culminating in treatment resistance. For this reason staging systems should to be formulated in parallel with treatment algorithms, as different phases have diverse therapeutic requirements. Therefore, it is assumed that staging is instrumental in choosing personalized optimal management plans, and is simultaneously helpful in elucidating illness prospects in individual cases. It must be clearly understood that early interventions are essential in preventing the diathesis from advancing and becoming treatment refractory. In this respect, while treatment during the initial stages should primarily focus on neuroprotective strategies, therapy during more advanced phases should be palliative and rehabilitative to control the consequences of disease progression. The importance assigned to neuroprotective approaches may take psychiatry to a new era of preventive medicine [69]. Figure 4 illustrates the basic tenets of management of mood disorders and is helpful in understanding the strategies required at early versus late stages (Figure 4).
LIMITATIONS OF STAGING MODELS
With respect to staging models for mood disorders the following caveats must be kept in consideration.
i. A developmental clinical staging approach incorporating childhood antecedents represents an important advance supporting earlier and more accurate diagnosis.
ii. Clinical staging of mood disorders must consider both the natural history and the heterogeneity of the diagnosis.
iii. A comprehensive evidence-based staging model is important to promote early detection and intervention, as well as identifying markers of illness predisposition and progression.
iv. The proponents of staging mood disorders on the basis of neuroprogression and allostatic load emphasize differentiating earlier from later phases in adult patients meeting full diagnostic criteria for a particular mood disorder.
v. These proposals neither consider the evidence of early antecedent risk factors nor the developmental illness trajectory, as reported in several large prospective studies of the offspring of parents with mood disorders.
vi. The likely staging models should be examined with the stipulation that these provide an aggregate view derived from research studies and will not apply to all high-risk offspring as environmental and epigenetic factors operate in the expression of the diathesis.
vii. The problem with Kapczinski’s allostatic load model is that it focuses solely on the progression of end-stage established disease, rather than presenting a paradigm based on the full natural history of illness evolution.
viii. While Berk et al.’s approach is more comprehensive, it collapses numerous discrete early clinical stages of illness development into a single very heterogeneous stage 1. Further, it does not incorporate family history as a way to identifying high-risk individuals, although a positive family history is the most important risk factor predicting for, at least, classical BD. Without the context of positive family history, the early stages are too nonspecific and would yield a very high rate of false positives.
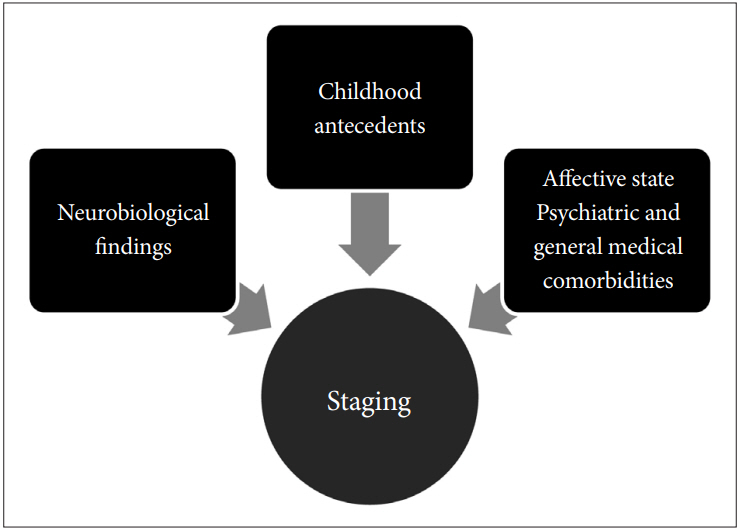
ix. Finally, to have applicable value the staging protocol must include early forerunners, and correlate the symptomatic state with existing comorbidities. Neuroimaging and peripheral biomarkers are corroborative factors that distinguish early from late stages [70]. Figure 5 is helpful in understanding the essential features of an adequate staging model and clarifies the prerequisites in this respect (Figure 5).
CONCLUSION
Staging of major psychiatric disorders is gaining momentum as the clinical significance of this framework is substantial. Several lines of research show that the course of principal mood disorders follows an expected and evolving path, from mild to progressively severer presentations. In view of this fact, a comprehensive staging model leads to a better understanding of mood disorders. While the blueprint of any staging system does not of necessity imply that patients would pass through each phase sequentially, it does give an overall view of how the illness unfolds as it develops. The extrapolative nature of the staging schema helps in formulating individually tailored treatment plans and defining prognosis. Appropriate and adequate treatment in the early phases should prevent advancement to the latter periods where lasting damage has, in essence, occurred. In susceptible individuals with an ultra-high risk profile, interventions in the earliest stages give a chance of preventing the disease from progressing and resulting in a whole host of undesirable consequences. Each one of the existing staging models has its merits and limitations, but unquestionably there is a great need for this proposal. It is expected that future research will lead to refinements in the staging protocols with benefit for the patients of mood disorders, which indeed are intractable conditions with poor prospects for many sufferers.