 |
 |
- Search
| Psychiatry Investig > Volume 16(6); 2019 > Article |
|
Abstract
To examine the feasibility of low-charge electrotherapy (LCE) in treating geriatric major depressive disorder (MDD) patients. Bi-temporal LCEs (approximately 25 mC) were performed with an electroconvulsive therapy (ECT) instrument three times per week. We used the Hamilton Depression Scale 17 (HAMD-17) and the Hamilton Anxiety Scale (HAMA) to assess the effects of LCE and the Mini-Mental State Examination (MMSE) to evaluate the cognitive function change before and after LCE. Six visits occurred at the baseline, after LCE sessions 3, 6, and 9, after the last session, and at the end of the one-month follow-up period. Four patients were enrolled in the study. Two patients completed all LCE sessions. Two patients withdrew during the trial, one due to the adverse event of uroschesis potentially caused by atropine and the other due to her own will. All four patients completed the follow-up sessions. The HAMD-17 and HAMA scores were reduced significantly at the last LCE session and the end of the follow-up period compared with the scores at the baseline. As measured by the MMSE, cognitive impairment showed no significant changes at the last LCE session and the end of the follow-up period compared with that at the baseline. In this case series, LCE showed potential as an alternative current-based treatment for treating geriatric MDD patients. Further research is needed to assess the efficiency and safety of LCE.
Geriatric major depressive disorder (MDD) is one of the most severe health problems in the world. As MDD causes a series of serious problems in elderly patients, rapid remission in geriatric MDD patients is important [1]. Routine first-line procedures, such as antidepressants or cognitive behavioral therapy (CBT), are insufficient for geriatric MDD patients; 55-81% of elderly patients fail to improve with first-line selective serotonin reuptake inhibitor (SSRI) or serotonin-norepinephrine reuptake inhibitor (SNRI) treatments. Electro-convulsive therapy (ECT), the most effective therapy for severe depression, has been used in clinical practice for decades. ECT has shown significant efficacy in geriatric MDD patients [2,3]. However, ECT also has a number of side effects, such as headache, delirium, forgetfulness, and cognitive impairment [1], which are especially severe among geriatric patients [4]. As a result, many elderly patients refuse ECT treatment, leading to a delay in the remission of depression and even loss of life. Some more recent procedures with fewer side effects, including repetitive transcranial magnetic stimulation (rTMS) and transcranial direct-current stimulation (tDCS), are used to treat MDD [1,5,6]. However, these new treatments are less effective than ECT [6-8]. Therefore, improving ECT to retain its therapeutic efficacy while minimizing its side effects will strongly benefit MDD patients.
After reviewing the literatures, we found an interesting phenomenon. Some ECT methods failing to induce seizures also demonstrated antidepressant effects but without severe side effects, such as cognitive impairment [9-12]. Notably, a recent open-label proof-of-concept study [13] demonstrated that low-charge nonconvulsive electrotherapy (NET) may have significant antidepressant efficacy. More importantly, the side effects of low-charge NET were moderate compared with those of ECT [13]. In summary, geriatric MDD patients may receive some significant benefits from these potential features of low-charge electrotherapy (LCE). Considering the sparse literature of the present field, we designed this case series as pilot study to examine the feasibility of treating geriatric MDD patients with LCE.
This case series was conducted in accordance with the latest version of the Declaration of Helsinki, and the Anhui Mental Health Center Research Ethics Committee approved our plan(2017-6). The inclusion criteria were as follows: 1) inpatient; 2) 60≤age≤80; 3) diagnosed with MDD according to the Diagnostic and Statistical Manual of Mental Disorders (DSM-5); 4) poor response to one month of SSRI/SNRI treatment; 5) current Hamilton Depression Scale 17 (HAMD-17) score≥24; 6) refused ECT; 7) voluntary participation in the study and 8) could sign informed consent form voluntarily. The exclusion criteria were as follows: 1) other comorbid mental disorders (i.e., bipolar disorder, psychotic disorders, and current substance abuse); 2) current suicidal ideas; 3) history of stroke, epilepsy or severe cardiovascular disease; and 4) history of allergy to anesthesia.
LCEs were performed with a Thymatron IV system integrated with an ECT instrument (Somatics, Lake Bluff, IL, USA) three times per week (Monday, Wednesday, and Friday). The percent energy dial was set to the minimum (5%, approximately 25 mC) with the DGX mode (Pulse width=1 ms; frequency=30 Hz). Patients were administered propofol anesthesia, and succinylcholine and atropine were used to relax the muscles and suppress glandular secretion during each treatment session. All patients underwent bi-temporal ECT electrode placement, and seizure activity was monitored via electroencephalography by the ECT instrument. The maximum number of LCE sessions was 12, and the patient could terminate the LCE procedure at any time.
Clinical status was measured at the baseline, every three LCE sessions, after the last LCE session and one month after the last LCE session. We used the HAMD-17 and Hamilton Anxiety Scale (HAMA) to assess depression status and anxiety level. The Mini-Mental State Examination (MMSE) was used to probe cognitive impairment, and any adverse event was recorded to analyze the safety of LCE.
We mainly performed descriptive statistics methods by using SPSS (IBM SPSS Statistics for Windows, version 21.0; IBM Corp., Armonk, NY, USA) to present the results of the study due to the small number of patients. An improvement in the HAMD score of>50% from baseline was considered a clinical response; a score of less than 7 was considered remission.
Four patients were enrolled in the case series and received LCE from May 01, 2017 to October 30, 2017. Two patients (Patients B and D) completed all LCE sessions. Ten convulsions were induced during the total 35 LCE treatments, the detailed information was showed in the supplemental digital content (Supplementary Table 1 in the online-only Data Supplement). Two patients (Patients A and C) withdrew during the trial. After five LCE sessions, Patient A was unsatisfied with the efficacy and withdrew. Patient C withdrew from the trial due to the adverse event of uroschesis potentially caused by atropine after the 8th LCE session, but both Patients A and C completed the follow-up sessions. We included the data from Patients A and C in the final analyses to avoid potential bias. There were declinations of more than 80% in the HAMD-17 and HAMA scores for the two patients who completed the LCE sessions (Patients B and D) after the last LCE session; the declinations in these scores for the two patients who did not complete the LCE sessions (Patients A and C) were more than 30%. However, the cognitive impairment measured by the MMSE showed no discernable change between the baseline and the last LCE session or the end of the follow-up period (Figure 1). Demographic characteristics are shown in Supplementary Table 1 (in the online-only Data Supplement); clinical and treatment characteristics are shown in Table 1.
To the best of our knowledge, the present study is the first to examine LCE in geriatric MDD patients. The patients in this trial achieved significant responses in terms of both depression and anxiety. Most notably, the cognitive function of all patients remained stable throughout and after the LCE procedures.
In the present case series, the mean decrease in the HAMD-17 score was approximately 60%, similar to other ECT trials as well as a NET study [13]. The mean decrease in the HAMA score was approximately 70%, similar to our former study and other ECT studies [14]. Most of the patients in the Regenold et al. [13] trial showed improvement, and our study further demonstrated that LCE is also effective in geriatric patients over the age of 60; these results suggested that LCE/NET may have antidepressant efficacy in all age groups of patients.
The mechanism of ECT’s antidepressant effect remains unclear. One hypothesis was that the current-induced seizures play a key role in the antidepressant effect. However, Regenold et al. [13] suggest that the current-induced seizures during ECT may be unnecessary. Furthermore, some other electricity-based treatments, such as rTMS and tDCS, which involve smaller currents without induced seizures, also exert certain effects on depression [1,5,6]. Although the effects of rTMS and tDCS are not as good as those of ECT [6-8], they have fewer side effects than ECT, meaning that the electric current, not the induced seizures, may be a key determining factor of efficacy. Another hypothesis was that the antidepressant effect is associated with the current path (i.e., electrode placement) [15]. Some studies have demonstrated a dose-response relationship between the antidepressant effect and the electric charges in right unilateral (RUL) ECT [15]. However, this dose-response relationship is absent in bi-frontal and bitemporal ECT. Regarding NET studies, the bi-frontal electrode placement employed by Regenold et al. [13] and the bitemporal electrode placement used in the present study both showed efficacy in MDD patients, meaning that the current path (electrode placement), not the absolute energy charge, might play a significant role in the higher current density observed in the prefrontal regions with the bi-frontal and bitemporal electrode placements [15]. However, this hypothesis requires further study. The diminution of currents during ECT, which could reduce the negative effects while maintaining the remarkable therapeutic effects, in geriatric patients will be a lasting topic of interest. Considering the potential benefits for geriatric MDD patients, our results demonstrate that LCE, or NET, should be further studied in the future [13,15].
The side effects of the present study are mild. One patient who reported urinary retention withdrew. This retention may be caused by atropine; however, no other severe side effects were observed during the LCE procedure. Furthermore, only one patient reported a single incident of a light post-LCE headache, which usually occurred after traditional ECT; she obtained relief from the headache soon after a nap. Regarding cognitive impairment, the differences in the MMSE score between the baseline and after LCE were not significant, which is consistent with NET [13]. This observation suggests that LCE is a reliable and safe antidepressant treatment. Although some studies have demonstrated improved cognitive function in elderly patients after complete ECT treatment or in the follow-up sessions [3,16], we noticed a decline in cognitive function during and a short time after ECT [16-20]. In our research, patients’ cognitive function remained stable during and at one month after the LCE procedures; we speculate that this stability may be related to the low currents involved in LCE. However, long-term (>one month) LCE cognitive changes should be studied in the future. Moreover, common comorbid diseases, such as cardiovascular disease, cerebrovascular disease, diabetes, and pulmonary disorders, in geriatric MDD patients may increase the risk of ECT [2]. In our opinion, LCE may reduce the risk of electrotherapy in elderly patients, and we anticipate more related studies.
Although the present case series study had several limitations, such as a small number of participants, an open-label design, and no controls, to our knowledge, it is the first LCE study on geriatric MDD patients. Further research is needed to probe the feasibility of LCE on geriatric MDD patients, and larger studies, especially randomized controlled studies, are needed to evaluate the efficacy and safety of this novel treatment.
Supplementary Materials
The online-only Data Supplement is available with this article at https://doi.org/10.30773/pi.2019.03.21.1.
ACKNOWLEDGEMENTS
This work was supported by the Natural Science Foundation of Anhui university major project (Grant No. KJ2018A0196), the Applied Medicine Research Project of Hefei Health and Family Planning Commission (Grant No. hwk2017yb015) and the Doctoral Foundation of the First Affiliated Hospital of Anhui Medical University (Grant No. 2017-1280).
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Xiao-ming Kong, Xin-hui Xie. Data curation: Yan Sun. Formal analysis: Xiao-ming Kong. Funding acquisition: Xiao-ming Kong, Yan Sun. Investigation: Hong Hong, Chen Wang. Methodology: Xiao-ming Kong, Yan Sun. Project administration: Xiao-ming Kong, Yan Sun. Supervision: Xiao-ming Kong. Visualization: Yang Chen. Writing—original draft: Xiao-ming Kong, Shu-xian Xu.
Figure 1.
A, B, and C show the change of the Hamilton Depression Scale-17, Hamilton Anxiety Scale, and Mini-Mental State Examination (MMSE) scores for each geriatric major depressive disorder patient before and after low-charge electrotherapy (LCE).
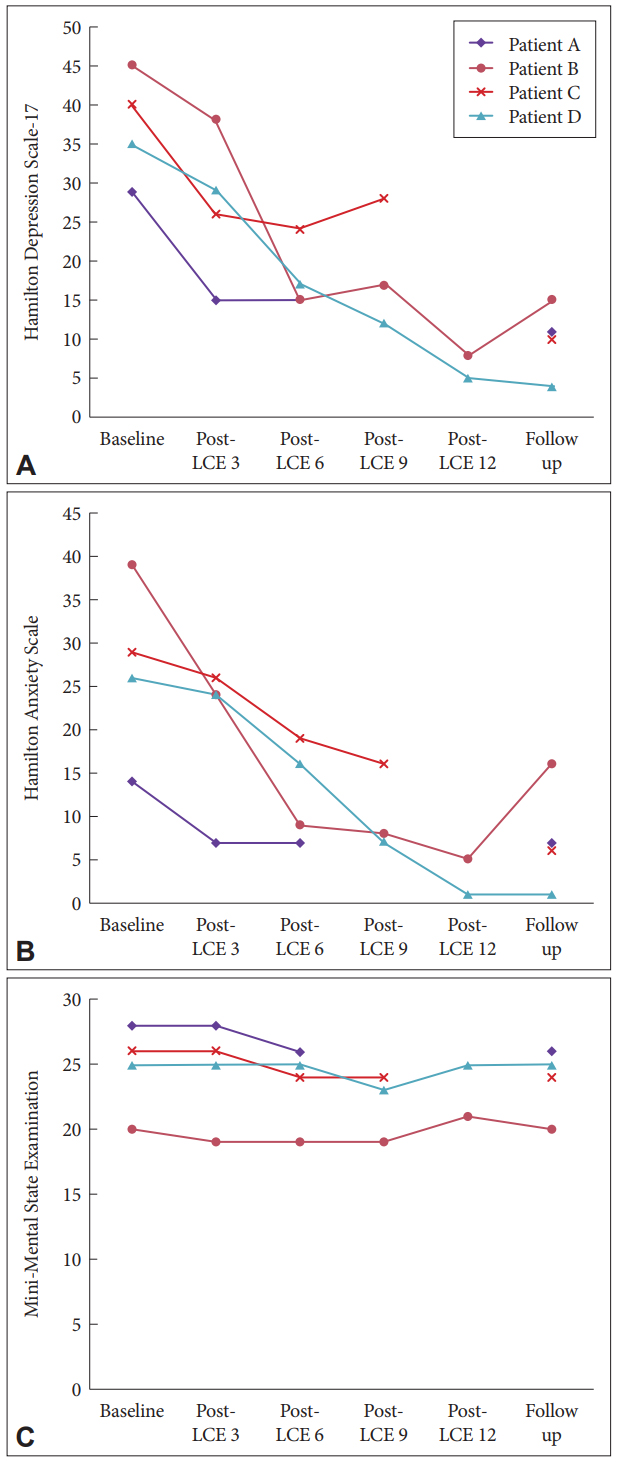
Table 1.
Detailed treatment characteristics
REFERENCES
1. Cusin C, Dougherty DD. Somatic therapies for treatment-resistant depression: ECT, TMS, VNS, DBS. Biol Mood Anxiety Disord 2012;2:14



2. Kerner N, Prudic J. Current electroconvulsive therapy practice and research in the geriatric population. Neuropsychiatry (London) 2014;4:33-54.



3. Hausner L, Damian M, Sartorius A, Frölich L. Efficacy and Cognitive Side Effects of Electroconvulsive Therapy (ECT) in Depressed Elderly Inpatients With Coexisting Mild Cognitive Impairment or Dementia. J Clin Psychiatry 2011;72:91-97.


4. McCall WV, Cohen W, Reboussin B, Lawton P. Pretreatment differences in specific symptoms and quality of life among depressed inpatients who do and do not receive electroconvulsive therapy: a hypothesis regarding why the elderly are more likely to receive ECT. J ECT 1999;15:193-201.


5. Loo CK, Alonzo A, Martin D, Mitchell PB, Galvez V, Sachdev P. Transcranial direct current stimulation for depression: 3-week, randomised, sham-controlled trial. Br J Psychiatry 2012;200:52-59.


6. Hansen PEB, Ravnkilde B, Videbech P, Clemmensen K, Sturlason R, Reiner M, et al. Low-frequency repetitive transcranial magnetic stimulation inferior to electroconvulsive therapy in treating depression. J ECT 2011;27:26-32.


7. Chen JJ, Zhao LB, Liu YY, Fan SH, Xie P, et al. Comparative efficacy and acceptability of electroconvulsive therapy versus repetitive transcranial magnetic stimulation for major depression: a systematic review and multiple-treatments meta-analysis. Behav Brain Res 2017;320:30-36.


8. Ren JJ, Li H, Palaniyappan L, Liu H, Wang J, Li C, et al. Repetitive transcranial magnetic stimulation versus electroconvulsive therapy for major depression: A systematic review and meta-analysis. Prog Neuropsychopharmacol Biol Psychiatry 2014;51:181-189.


9. Alexander L. Nonconvulsive electric stimulation therapy; its place in the treatment of affective disorders, with notes on the reciprocal relationship of anxiety and depression. Am J Psychiatry 1950;107:241-250.


10. Beran M, Perkins JC, Scollon RW. Psychological studies on patients undergoing nonconvulsive electric-stimulation treatment. Am J Psychiatry 1952;109:367-374.


12. Prudic J, Sackeim HA, Devanand DP, Krueger RB, Settembrino JM. Acute cognitive effects of subconvulsive electrical stimulation. Convuls Ther 1994;10:4-24.

13. Regenold WT, Noorani RJ, Piez D, Patel P. Nonconvulsive electrotherapy for treatment resistant unipolar and bipolar major depressive disorder: a proof-of-concept trial. Brain Stimul 2015;8:855-861.


14. Kong XM, Xu SX, Sun Y, Wang KY, Wang C, Zhang J, et al. Electroconvulsive therapy changes the regional resting state function measured by regional homogeneity (ReHo) and amplitude of low frequency fluctuations (ALFF) in elderly major depressive disorder patients: an exploratory study. Psychiatry Res Neuroimaging 2017;264:13-21.


15. Sackeim HA. Is the seizure an unnecessary component of electroconvulsive therapy? A startling possibility. Brain Stimul 2015;8:851-854.


16. Dybedal GS, Tanum L, Sundet K, Bjølseth TM. The role of baseline cognitive function in the neurocognitive effects of electroconvulsive therapy in depressed elderly patients. Clin Neuropsychol 2015;29:487-508.


17. Semkovska M, McLoughlin DM. Objective cognitive performance associated with electroconvulsive therapy for depression: a systematic review and meta-analysis. Biol Psychiatry 2010;68:568-577.


18. Bodnar A, Krzywotulski M, Lewandowska A, Chlopocka-Wozniak M, Bartkowska-Sniatkowska A, Michalak M, et al. Electroconvulsive therapy and cognitive functions in treatment-resistant depression. World J Biol Psychiatry 2016;17:159-164.


- TOOLS
-
METRICS

-
- 1 Crossref
- Scopus
- 5,204 View
- 82 Download
-
The Development of the Korean Medication Algorithm for Major Depressive Disorder2005 September;2(2)






