Disrupted Control Network Connectivity in Abstinent Patients with Alcohol Dependence
Article information
Abstract
Objective
Alcohol causes damage to the brain and is associated with various functional impairments. However, much of the brain damage can be reversed by abstaining for enough time. This study aims to investigate the patterns and degrees of brain function in abstinent patients with alcohol dependence by using resting-state functional connectivity.
Methods
26 male patients with alcohol dependence (alcohol group) and 28 age-matched male healthy volunteers (control group) were recruited from a mental hospital and the community, respectively. Using 3T MRI scan data, the resting-state functional connectivity of the task-negative and task-positive networks was determined and compared between the groups.
Results
There were no significant group differences in the resting-state functional connectivity in the default mode or in the salience and sensorimotor networks. Compared with the control group, the alcohol group showed significantly lower functional connectivity in the executive control network, especially in the cingulo-opercular network and, in some regions of interest, the dorsal attention network.
Conclusion
This finding suggests that some brain networks do not normalize their functions after abstinence from drinking, and these results may be helpful in future research to investigate the mechanisms for craving alcohol and alcohol relapse prevention.
INTRODUCTION
Problem drinking, like alcoholism, causes structural changes and functional consequences in the human brain and progresses to variable mental symptoms, including cognitive decline. These brain changes are not consistent in each individual and range widely from slight nervous damage to severe neurological consequences, including Wernicke encephalopathy (WE) and Korsakoff's syndrome (KS). These mechanisms of irreversible cognitive disorders or mental disorders include ethanol-specific effects, thiamine deficiency, liver dysfunction, and synergistic effects.1
Although it is obvious that heavy drinking is related to brain damage, there are mixed opinions about the effects of mild to moderate drinking on the human body and brain. At the same time, the recovery potential and the rate of alcohol-induced brain damage after short- and long-term abstaining is diverse and unclear. Many studies used clinical outcome variables such as neuropsychological testing23 or structural imaging such as white matter damage4 in order to determine the effect of mild to moderate drinking or abstaining on brain function after heavy drinking.
Resting-state functional connectivity magnetic resonance imaging (rs-fcMRI) is a useful and sensitive method for assessing neuronal activity and brain function.5 The baseline blood oxygen level-dependent (BOLD) activity is topologically organized in multiple spatially distributed resting-state networks (RSNs).6 The rs-fcMRI technique can determine functional connectivity by assessing the temporal correlation of spontaneous fluctuations of the BOLD signal between regions. The default mode network (DMN), as the classic task negative network, has been the main network of interest innumerous research studies of Alzheimer's disease or normal aging.78 At the same time, there is a growing interest in the resting-state task-positive network, which activates in cognitively demanding settings and shows anticorrelation with the task-negative network.910
The rs-fcMRI was used to evaluate brain function in not only normal aging or dementia disorders but also many neuropsychiatric disorders such as multiple sclerosis, depressive disorders, schizophrenia, attention-deficit hyperactivity disorder, and autism.511 Furthermore, addictive disorders such as alcoholism are good targets for using structural and functional imaging studies. For example, brain atrophy related to chronic drinking could be partially recovered with abstinence, and the rate of recovery happens more quickly in complete abstinence compared to continuing a small amount of drinking.12 There was subsequent atrophy in the areas associated with error monitoring and behavior control and increases of brain activity and connectivity in the areas related to processing salient or aversive stimuli in relapsed patients with alcohol dependence.13
Many of the studies that identified resting-state network changes in alcohol-related disorders showed mixed results, such as a disruption of connectivity of the DMN14 and reward processing network and increasing synchrony of the executive control network.15 However, the results of these studies were not consistent because clinical characteristics of alcoholics were heterogeneous, and the definitions of RSNs they used were not identical across the studies. In addition, both the decrement of the RSN and the compensatory neural ability and dedifferentiation were observed in the brains of alcoholics.16 Therefore, it is necessary to explore alcoholics' connectivity in systematic, well-classified task-negative and task-positive networks in order to expand our understanding of the effects of alcohol consumption and abstinence on the human brain network.
Although there may be confusion in the demarcation and overlap of the structures belonging to specific RSNs across the studies, many task-negative and task-positive networks, such as the cognitive control network (CON), the dorsal attention network (DAN) and the salience (SAL) network, were identified. In a comprehensive paper review, Brier and colleagues classified the many resting-state networks such as the DMN, the DAN, the CON, the SAL, and the sensorimotor network (SMN), and they suggested coordinates for each network.17
We assessed the brain functions in previously RSN settings of abstinent patients with alcohol dependence by using rs-fcMRI, and we compared their functions to that of social drinkers to ascertain the effects of alcohol consumption and abstinence on the brain functions of patients with alcohol dependence.
METHODS
Participants
Participants included male patients admitted to a mental hospital specializing in alcohol dependence in Cheongju, South Korea, from March 1, 2014, to August 31, 2014. In addition, age-matched healthy controls ranging from 40 to 65 years of age were recruited through an advertisement. The diagnosis of alcohol dependence according to the 4th Edition of Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) was confirmed using structured interviews (SCID).18 All participants had received ongoing treatment for alcohol use disorder (AUD) for a minimum of three months with complete abstinence. Their mean abstinence period was 13.2±16.1 months. Participants who had a major mental disorder, neurologic disease, severe systemic disease, or other substance use disorder, except nicotine and caffeine use disorders, were excluded. This process left us with 26 patients with alcohol dependence and 28 healthy controls. All processes of this study were approved by the Bioethics Committee Board of Chungbuk National University. All participants gave written informed consent and their anonymity was preserved.
MRI acquisition
All MRI images were collected on a 3T Philips Achieva scanner (Philips Medical System, Best, Netherlands) at the Ochang campus of the Korean Basic Science Institute. All participants were scanned by the same 32-channel head coil and the same pulse sequence. They were instructed to lie still in relaxed state and keep their eyes fixating while remaining awake.
Volumetric magnetization-prepared rapid gradient echo (MPRAGE) parameters were referenced from the Alzheimer's Disease Neuroimaging Initiative (ADNI) protocol [TR=6.8 ms, TE=3.1 ms, flip angle=9 degrees, 256×256 (1×1 mm) inplane resolution, 170 1.2-mm slices without gaps].
The gradient spin echo sequence for the resting state functional imaging was used (TE=30 ms, TR=3000 ms, field of view=212 mm, flip angle=80 degrees, 3 mm isotropic voxels). Two scans for 7 minutes (140 volumes per scan) were performed during the participants' resting state.
Image processing
We used conventional methods for the connectivity analysis with an in-house script consisting of AFNI, FSL, and Free-Surfer commands.1920 This step included compensation for slice-dependent time shifts, elimination of systematic odd-even slice intensity differences due to interleaved acquisition, and concatenation of the BOLD volumetric time series. After that, we aligned the concatenated echo-planar imaging (EPI) data to the MPRAGE imaging registered to a Montreal Neurological Institute (MNI) space.
After normalizing the signal to a percent signal change, several additional preprocessing steps were utilized to reduce any spurious variance that was unlikely to reflect neuronal activity.21 These steps included 1) temporal filtering retaining frequencies in the 0.009–0.08 Hz band; 2) removal by regression of several sources of variance, such as i) regression of six parameters obtained by rigid body head motion correction, ii) regression of the whole brain signal averaged across the whole brain, iii) regression of ventricular signal averaged from ventricular regions of interest (ROI), iv) regression of white matter signal averaged from white matter ROIs, and v) regression of first derivatives of the above regressors; and 3) spatial smoothing (6 mm full width at half-maximum Gaussian blur).
We also performed the motion scrubbing procedure suggested by Power and colleagues to improve reliability. This procedure censors and removes unreliable time series by estimating frame displacement (FD) and DVARS (D referring to the temporal derivative of time course, VARS referring to the RMS variance over voxels).22
Rs-fcMRI procedures
Thirty-six spherical (6 mm radius) ROIs, originally representing five RSNs, were derived by reviewing related papers.17 These canonical RSNs-DMN, DAN, CON, SAL, and SMN-correspond to critical brain functions. Correlation maps were produced by extracting the time courses from each seed region. Time courses from the same bilateral structures were averaged to one value. Therefore, time courses representing 21 areas were extracted to further the process. The Pearson correlation coefficient (r) was computed between the time courses of the 21 areas. Statistical tests of the rs-fcMRI results were computed after application of Fisher's z transformation {z=0.5 ln [(1+r)/(1-r)]}. We used the individual z scores representing connectivity between two ROIs and the averaged values of the z scores in all ROI pairs composed of each RSN as an intra-network composite score representing overall intra-network connectivity to compare group differences.
Because of the relatively small numbers of areas in CON, considering the assumption of its major role in alcoholics' brains, we searched the more ROIs belonging to the CON classification by their functional difference. Finally, we estimated the connectivity of the fronto-parietal task control network (FPTC), consisting of 25 ROIs, and the cingulo-opercular task control network (COTC), consisting of 14 ROIs, with the same method. Their ROI coordinates were referred from the Power ROIs (http://www.nil.wustl.edu/labs/petersen/Resources_files/Consensus264.xls).23
Statistical analyses
Analyses were performed using the CRAN R statistical package, version 3.1.3 (http://r-project.org). Participant characteristics were examined using Student's t-test and nonparametric statistics, where appropriate. The differences in the resting state functional connectivity between the groups were tested with Student's t-test, and the effect size of group differences were estimated using Cohen's d value.
Scatter plots were examined and tests for linear relationships were conducted using multivariable hierarchical linear regression models. Age as a continuous variable was centered prior to analysis. The following regression model was used for the final analyses of functional connectivity, testing the null hypothesis: βi=0: Y=β0+β1*(age)+β2*(group, alcohol group=1, control group=0)+ε, where Y is the observed functional connectivity for the each network.
RESULTS
Participants' demographics
All 54 participants (26 in the alcohol group, 28 in the control group) were men aged 40–65. There were no differences in age distribution, education level, or socioeconomic status between the groups. However, the mean weight of the control group was higher than that of the alcohol group. In addition, there were more divorced persons in the alcohol group. Detailed demographics are shown in Table 1.
Comparison of connectivity between groups
The intra-network connectivity of the alcohol group was significantly lower than the connectivity of the control group in the CON (t=-2.05, p<0.05, Cohen's d=0.57). However, there were no group differences for the intra-network connectivity in the DMN, DAN, SAL, and SMN. After comparing the group differences of individual connectivity between the one seed region and other spheres composed of each network, we found there were significant group differences between medial temporal complex (MT) and posterior intraparietal sulcus (pIPS), anterior intraparietal sulcus (aIPS) within DAN, dorsomedial prefrontal cortex (dmPFC) and superior parietal (SP) within CON (Table 2). ROI pair correlation maps are shown in Figure 1.

ROI pair correlation matrices for alcohol group (A) and control group (B). The ROIs are grouped by RSN (A-1 and B-1, DMN, DAN, CON, SAL and SMN, respectively). Intranetwork correlations appear on diagonal blocks. The COTC ROI pair correlation matrices were shown in A-2 and B-2. Units are z-transformed correlation coefficients. ROI: region of interest, RSN: resting state network, DMN: default mode network, DAN: dorsal attention network, CON: control network, SAL: salience network, SMN: sensorimotor network, COTC: cingulo-opercular task control network.
Multiple regression models
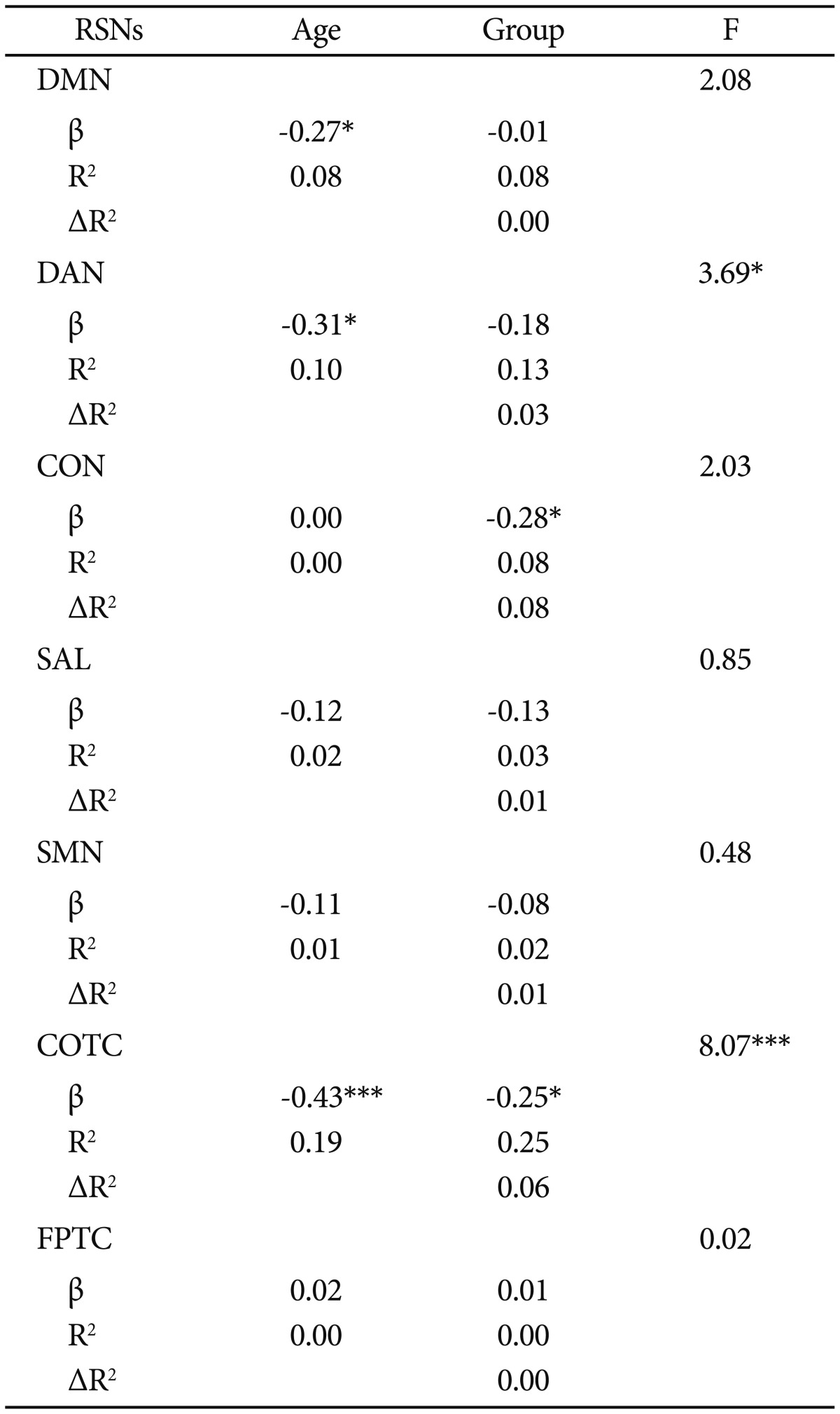
The multiple hierarchical regression model, including age and group as covariates and connectivity as a dependent value, showed no valid model in DMN, DAN, CON, SAL, and SMN, but there was a valid regression model in COTC. Overall, the connectivity of COTC was inversely associated with age, accounting for 19% of the unique variance (p<0.001), and the alcohol group was associated with less connectivity, accounting for 6% of the unique variance (p<0.05) (Table 3).
DISCUSSION
The characteristic individual of the alcohol group in this study had drank 28,941 units in his lifetime and had abstained for at least three months. This study tried to assess the effect of abstinence on the pattern of functional recovery from brain damage caused by long-time alcohol consumption.
Many studies based on a family history of alcoholism have shown impairments of brain activities and cognitive functions related to task-positive networks, such as decision making, working memory, spatial memory, reaction suppression, and attention prior to beginning drinking.2425 These results are consistent with the opinion that the risk of alcoholism is transmitted genetically. Therefore, the differences of connectivity shown in this study could have been induced not by the effect of long-term alcohol drinking but by genetic transmission. To overcome the blind spots of this cross-sectional study, which cannot prove the cause-and-effect relationship, a longitudinal follow-up study is needed. Nevertheless, chronic alcohol consumption induces brain damage and cognitive impairment and causes clinical situations such as alcohol-related dementia, WE, and KS, and their neuropathologic and neuroimaging findings are well known.1 It is valuable to ascertain the brain functions of long-term abstainers with alcohol dependence after the experience of long-term drinking.
Major group differences in this study focused on specific task-positive networks, mainly the CON and the DAN. Specifically, a group difference of connectivity between the dmPFC and the SP within the CON was prominent, and a group difference of connectivity between the MT and the aIPS, the MT and the pIPS within the DAN was also observed. However, we could not observe any significant group differences of connectivity among areas composed of the DMN, the SAL, and the SMN.
For now, the most interesting network in studies of brain connectivity is the classic DMN. There was a report of the impairment of connectivity in the network of patients with alcohol dependence.14 However, the findings could not cover whole areas of the networks and was limited to connectivity from only the left posterior cingulate cortex (PCC), and they did not find any significant group differences of connectivity between the right PCC and other areas composed of the DMN. We could not find significant differences in the whole connectivity of the DMN assessed by calculating the composite index and the specific individual connectivity among areas composed of DMNs, including the PCC and the cerebellum. Unlike their study,14 the single midline PCC was used as the seed region in this study. The different results between studies could have originated from the differences in the sample size or abstinence periods. The sample size was greater and the mean abstinence period was longer by approximately six times in this study.
We could not find the group differences of connectivity in the SAL. The SAL is composed of the paralimbic area and is related to internal emotions, such as anxiety, pain, and reactions to the autonomic nervous system. Meanwhile, the executive CON is known to be part of the dorsal neocortical executive control system and is related to executive function.1013 Our results provide the possibility and biological base that emotional recovery could occur before cognition recovery, like executive function in patients with alcohol dependence who abstained for more than three months after persistent problem drinking.
We found significant group differences of connectivity in the DAN and the CON. These networks belong to the taskpositive network. Ridderinkhof and colleagues classified the dorsolateral prefrontal cortex, ventrolateral prefrontal cortex, anterior cingulate cortex, supplementary motor area (SMA), preSMA, premotor cortex, motor cortex, thalamus, striatum, insula, cerebellum, and several other areas as part of the executive cognitive control network,24 but researchers' classifications are not identical. However, many studies have suggested the inefficient hyperactivity of the executive control network in patients with alcohol or other drug dependency or the connectivity decrement of the circuit's resting state.2627 The impairment of top-down regulation induced by the connectivity decrement of the executive control network could be related to cognitive impairment or relapse in patients with alcohol dependence.
Meanwhile, intrinsic connectivity networks that involve task-positive regions include the COTC, the FPTC, and the DAN.28 The DAN is proposed to underlie selective attention, especially in visual and spatial domains;929 the FPTC is thought to involve adapting after errors associated with the phasic aspects of attentional control; and the COTC is related to stable maintenance of task mode and strategy.3031 Furthermore, there is evidence that tonic alertness associated with vigilance or sustained attention is a fundamental function of the COTC.28
Disruption of connectivity in the DAN and the COTC shown in this study may provide the biologic base that recovery from impairment of long-term or sustained drinking could not be achieved in spite of abstinence for long periods. Considering the report that an avoidant reaction to arousal is related to craving and that subjective craving is important as a correlate of attention biases in abstinent alcoholics,32 we can infer the relationship between the COTC connectivity and clinical variables such as craving decrement or relapse prevention. Camchong and colleagues focused on the reward circuit and the executive control system of the abstinent patients and showed the association between the connectivity of these circuits and abstinence maintenance.15
We also need to consider several limitations. Although we ascertained the connectivity among every ROI sphere, we compared the group differences of connectivity by averaging the composite values as the representative value of each RSN. Some group differences between specific areas were hidden because averaged values were used in this study. However, the seed areas were drawn from reliable previous works, and the disruption of intra-network connectivity was shown in the brain of normal aging or Alzheimer's disease, which can develop dedifferentiation. Therefore, the overall intra-network disruption of the CON, the COTC, and the DAN in the alcohol group is meaningful.
Individuals in the alcohol group participated at least three months after abstinence. The results in this study were a mixture of brain damage from past drinking and the ability to recover from abstinence. However, individual differences in recovery ability from brain damage were not considered, so this is one of our limitations. The factors for the ability to recover from brain damage such as the BDNF genotype should be considered for future research.32 The results of the neurocognitive test should be matched to imaging data, but we were not able to include these data in this study. Therefore, these findings were limited to matching the group differences in connectivity with cognitive errors shown in alcohol dependence in previous studies.
The other main limitation of this study is the effect of psychotropic medications because of the inpatient setting for the alcohol group. All participants in the alcohol group took anticraving agents or benzodiazepines, so it was impossible to exclude the drug effect completely. However, a recent report that assessed the function of the executive network of alcohol use disorder did not show any differences in connectivity before and after the use of lorazepam or disulfiram.33
Despite these limitations, this study's strength is that high-quality neuroimaging data for a significant number of a focused and consistent population, like middle-aged males, were acquired to compare brain function. Based on these data, future studies should be done to research the genetic association, cognitive impairment, craving, and the relapse mechanism of alcohol dependence.