Asymmetrical Electroencephalographic Change of Human Brain During Sleep Onset Period
Article information
Abstract
Objective
Human cerebral hemisphere is known to function asymmetrically with daytime left hemisphere superiority in most right-handed persons. It may have relevance to the localization of specific function of the brain. This study attempted to reveal whether the functional cerebral asymmetry in the wakeful state is still maintained throughout the sleep onset period.
Methods
Thirty-channel EEG was recorded in 61 healthy subjects. The EEG power spectra of each of the seven frequencies were compared between the two kinds of 30-second states; the wakeful stage and the late-sleep stage 1.
Results
The asymmetrical indices of sleep stage 1 at several fronto-central leads were decreased in the delta, theta, alpha-2, and all beta bands. Conversely, at parts of parieto-occipital leads showed an increase in the indices of the theta, alphas, beta-1, and beta-2 bands. Any fronto-central leads did not show an increase in the index, and no parieto-occipital leads showed a decrease.
Conclusion
During the sleep onset period, power spectral asymmetry of the brain showed a different pattern from the wakeful stage. This asymmetrical pattern of EEG powers may suggest a reversal of the left hemispheric dominance during sleep.
INTRODUCTION
The human brain consists of two hemispheres, and they have been known not to be completely symmetrical with respect to their structural or functional aspects. Structural asymmetry of the brain was reported in the greater left planum temporale,12 longer left Sylvian fissure,2 and greater folding of left frontal operculum,3 as well as the superior parietal lobules and angular gyri.4 These left-right asymmetries are seen not only in humans, but also in the other greater apes and even in rodents.5
The hemispheric asymmetry was also found in their function, in addition to their structure. In an electrophysiological ACCESSstudy, Benca et al.6 reported frontal alpha asymmetry, and a functional brain imaging study by Swanson et al.7 also described functional asymmetry of the default mode network.
During wakefulness, an electroencephalographic (EEG) study observed a greater left frontal absolute power and a greater right occipital delta power compared with the other side.8 The other study with fifty-nine healthy subjects reported right side dominance of alpha oscillation on the fronto-central area and vice versa on the parieto-occipital area.9 The majority of reported investigations regarding resting EEG asymmetry is about frontal alpha asymmetry, and these studies have suggested that it is the correlates of emotional regulation and flexibility,1011 or stress response and cortisol levels.12131415
Regarding the sleep state, a unilateral bright-light exposure has been shown to affect EEG asymmetry during sleep,16 and Casagrande and Bertini demonstrated a re-patterning of the laterality across wake-sleep state using the behavioral method.17
These cerebral asymmetries assign a greater dominance to one specific hemisphere–especially to the left side–according to motor and language functions. The left hemisphere is responsible for language functions, such as sentence construction, word fluency, while the right hemisphere is responsible for visuo-spatial attention.181920
The purpose of this study is to investigate how electrophysiological appearance of cerebral asymmetry changes during the sleep onset period relative to the arousal state.
METHODS
Subjects
Sixty-one subjects with normal health status were recruited at a university hospital in Korea; there were thirty-four males with an average age of 26.6±2.4 years, with a range of 23–34 years, and twenty-seven females with an average age of 28.0± 3.5 years, with a range of 23–35 years. All subjects were normal, healthy, and right-handed with normal EEG findings. Subjects were excluded from the study if they had any a history or current state of neurological disorders, psychiatric illnesses, cardiovascular diseases, pulmonary disorders, substance abuse, obstructive sleep apnea syndrome, and primary or secondary sleep disorders. Subjects with a history of alcohol consumption within 24 hours before the test were also excluded. The unreported depressive or anxiety symptoms of subjects were evaluated with Beck Depression Inventory (BDI) and Beck Anxiety Inventory (BAI),2122 and subjects with BDI scores of greater than 13 or BAI scores of greater than 15 were excluded. We also excluded subjects who met the criteria for obesity classes I, II, and III, as defined by the World Health Organization, with a body mass index of more than 29 kg/m2.
Subjects were instructed to perform sleep hygiene for a week prior to the EEG. Written consent was obtained from each participant. The study protocol was approved by the local Institutional Review Board (KUH1050056).
Electroencephalography
EEG measurements were taken between 10 am and 11 am by Neuroscan Version 4.3.3 (Compumedics, Abbotsford, Victoria, Australia). The international 10±20 system with 32 channels were used, and the reference leads were placed in the ears on A1 and A2 sites. The laboratory was electrically shielded, protected against noise, and equipped with temperature and humidity control systems.
To eliminate the artifacts that possibly arise from the movements of the eyeball or eyelid, we put horizontal electro-oculography channels at 1-cm away from the outer portion of each eye and vertical electro-oculography channels above and below the left eye, and we also used a correction software embedded in Neuroscan. The EEG acquisition parameters were a sampling rate of 500 Hz/channel, a high-pass filter of 0.05 Hz, a low-pass filter of 60 Hz, and a notch filter of 60 Hz.
The 30-second EEG measurement was performed on subjects with semi-supine positions and both eyes were closed. Arousal (the wakeful state) and sleep state (the late stage 1 sleep) was distinguished by the distribution of the alpha and theta waves in O1 and O2 electrodes. The wakeful state was determined as the first 30-second epoch exhibiting a strong alpha pattern for the whole period. The late stage 1 sleep was defined as the final 30 seconds of stage 1 sleep.
Statistical analysis
Brain waves for the analysis were delta (1–3.5 Hz), theta (4–7.5 Hz), alpha-1 (8–10 Hz), alpha-2 (10–12 Hz), beta-1 (12.5– 18 Hz), beta-2 (18.5–21 Hz), and beta-3 (21.5–30 Hz) bands. The power spectrum was calculated for 30 seconds of the time series data, and the asymmetry index was defined as log10 (the power of the left electrode)-log10 (the power of the right electrode). Statistical comparisons of the asymmetry index were performed between the two states for twelve electrode pairs (Fp1–Fp2, F3–F4, F7–F8, FC3–FC4, FT7–FT8, C3–4, T7–8, CP3–4, TP7–TP8, P3–P4, P7–P8, and O1–O2), and a statistical significance was set at p<0.01.
RESULTS
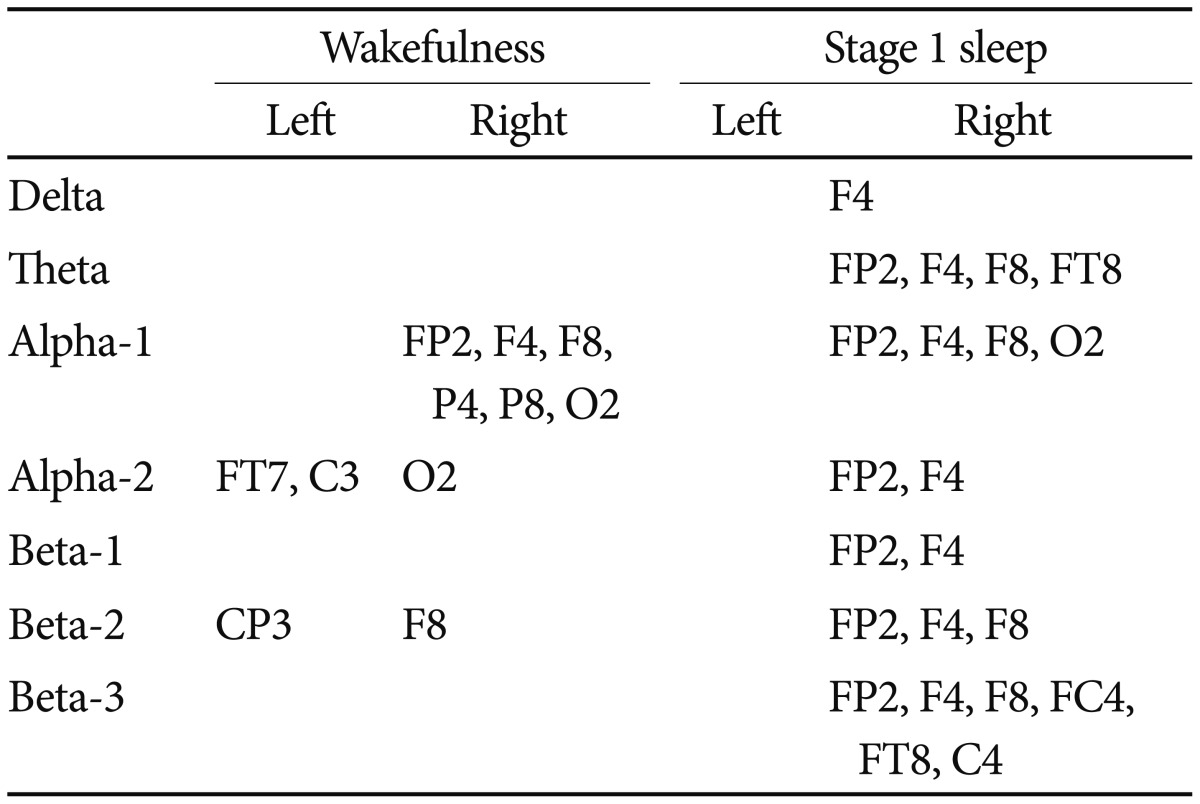
Left-right power difference (Table 1)
In the wakeful state, the left hemisphere showed a greater power for alpha-2 at the FT7 and C3 electrodes and for beta-2 at the CP3 electrode. Conversely, the right hemisphere showed predominance for alpha-1 at the FP2, F4, F8, P4, P8, and O2 electrodes, for alpha-2 at the O2 electrode, and for beta-2 at the F8 electrode.
In the late stage 1 sleep, the right hemisphere showed predominance for all EEG frequencies (delta at F4, theta at FP2, F4, F8, FT8, alpha-1 at FP2, F4, F8, O2, alpha-2 at FP2, F4, beta-1 at FP2, F4, beta-2 at FP2, F4, F8, beta-3 at FP2, F4, F8, FC4, FT8, and C4). No left hemispheric electrodes showed greater power than the right ones.
Left-right and sleep state interaction (Figure 1)

The change of asymmetric indices in the wakeful state and late-sleep stage 1. The indices decreased in the late-sleep stage 1 compare to the wakeful state except the indices of beta-2 wave in O1/2 lead and beta-1 wave in P3/4 lead (statistics by paired t-test, p<0.01).
As consciousness changes from the wakeful state to the late stage 1 sleep, the asymmetry index of beta-1 at the P3/4 electrode and of beta-2 at the O1/2 electrode increased, but, were reduced for delta at the CP3/4 electrode, for theta at the FP1/2 electrode, for alpha-2 at the FT7/8, C3/4, and CP3/4 electrodes, for beta-1 at the T7/8 electrode, and for beta-3 at the CP3/4 electrode.
DISCUSSION
Functional asymmetry of the human brain is prominent in handedness and language functions,2324 and these functions tend to exhibit the dominance of the left hemisphere. However, the asymmetry of the brain is derived not only from those two functions. Cerebral asymmetry is a phenomenon not only in humans but also in other primates. In addition to the handedness and language function, there is also asymmetry in discriminating faces and recognizing sounds other than language.252627 This asymmetry has been reported to differ between men and women.282930 It has been found that the surface area of the corpus callosum compared to the brain volume is gradually smaller as it gets closer to human in primates, which is an important fact about the cause of brain asymmetry.31
In other words, as the amount of information exchanged between the right and left halves increases, the amount of brain volume must increase. Due to the anatomical limitations, the amount of information exchanged in the right and left halves cannot be increased without increasing the total brain capacity. Therefore minimizing the amount of information exchanged on the left and right is advantageous in allocating brain capacity to increase local brain function. This is why not only the functional differences in the right and left hemispheres but also the difference between the anterior and posterior parts of the brain must exist.
These asymmetries of the brain are, of course, reflected in functional brain imagings or various neuropsychological tests. Neuropsysiological indicators such as brain waves can, of course, reflect those. Most investigations about EEG asymmetry have studied frontal alpha rhythms. The meaning of alpha rhythm is still controversial, however the hypothesis holding that increased alpha power means the inactivity of the local cortical area was widely supported by various findings, such as increased alpha powers correlated with a decreased BOLD signal in brain areas,32 posterior alpha increases on eyes closed condition33 and motor cortical alpha increases with limbs at rest.34 Functionally, frontal alpha asymmetry have been suggested to be involved in emotional regulation or flexibility in response to psychological stress regardless of state or trait dependency.1112 In that cases, left hemispheric dominance usually meant positive affective functioning.
On the transition from awakening to sleep, all parts of the brain do not fall asleep at the same time. There are some reports that the sleep proceeds to the posterior from the anterior of the brain,3536 and other report that sleep onset process have different patterns according to the waveform of brain waves and cortical areas.37 Several reports suggested that the laterality of human brain can be changed or reversed during sleep. By behavioral method using finger tapping test, Casagrande and Bertini found the right side had short inter-tapping interval (ISI) in wakeful condition whereas the left hand showed shorter ISI during NREM and REM sleep.17 Left-right EEG asymmetry of the beta/delta ratio was reported to gradually reduced after sleep onset.38 Some studies found the right hemispheric power becoming predominated except beta,394041 and two of these studies also reported a decrease in interhemispheric coherence during SOP.4041
In this study, frontotemporal (FT7) and centroparietal (C3, CP3) electrodes had left side predominance in wakeful state, whereas right frontal, parietal and occipital electrodes became superior to the left side in sleep state. As for changes of the left-right asymmetry index, posteromedial beta oscillation showed increasing pattern, but most frontal, frontotemporal and central electrodes became more right predominance patterns during the sleep onset period. In summary, no left electrode was superior to the right in sleep state, and left-right asymmetry indices from five out of twelve electrode pairs decreased in five out of seven frequency bands.
The reason why the laterality is different at the time of awakening and sleeping is not known precisely, but several hypotheses are known. The first is the so-called “Homeostatic hypothesis.” This hypothesis is based on the restorative function of sleep, which is regarded as one of the most important functions of sleep. Because the dominant hemisphere has much more information and stress to be processed when being awake than the non-dominant hemisphere has, it should take more rest during sleep and this seems to make non-dominant hemispheres look relatively more active.1742
The second hypothesis focuses on the intrinsic function of the non-dominant hemisphere itself and is also called the “Arousal hypothesis.” It is known that the right hemisphere more contributes to the attention or sustained vigilance than the left hemisphere.1743 In the sleep, the right hemisphere can be relatively dominant because the vigilance needs to be maintained somewhat relative to other functions.17
In this study, asymmetry index generally changed toward the right hemisphere dominant pattern during SOP. However, beta waves at the posterior medial electrodes showed different directions toward the left hemisphere. Despite the various limitations such as any possibility of the different SOP pattern between man and woman, though we can hardly find articles about it, and the possibility of hidden artifacts from relatively long recording time, the meaning of this study attributes to revealing the different asymmetry changing pattern of posterior medial beta waves from other parts of the brain during SOP with relatively many electrode pairs for all the usual bandwidth of EEG. However, further studies on the meaning and function of posterior beta waves are required to completely understand the reversal phenomenon of asymmetry.
Acknowledgments
This work was supported by the intramural research grant of Chungbuk National University in 2015.