Association Between Vitamin D Insufficiency and Metabolic Syndrome in Patients With Psychotic Disorders
Article information
Abstract
Objective
This study examined the association between vitamin D and metabolic syndrome in patients with psychotic disorders.
Methods
The study enrolled 302 community-dwelling patients with psychotic disorders. Sociodemographic and clinical characteristics, including blood pressure, physical activity, and dietary habit were gathered. Laboratory examinations included vitamin D, lipid profile, fasting plasma glucose, HbA1c, liver function, and renal function. Vitamin D insufficiency was defined as <20 ng/mL. Clinical characteristics associated with vitamin D insufficiency were identified.
Results
Among the 302 participants, 236 patients (78.1%) had a vitamin D insufficiency and 97 (32.1%) had metabolic syndrome. Vitamin D insufficiency was significantly associated with the presence of metabolic syndrome (p=0.006) and hypertension (p=0.017). Significant increases in triglycerides and alanine transaminase were observed in the group with a vitamin D insufficiency (p=0.002 and 0.011, respectively). After adjusting for physical activity and dietary habit scores, vitamin D insufficiency remained significantly associated with metabolic syndrome and hypertension.
Conclusion
Vitamin D insufficiency was associated with metabolic syndrome and was particularly associated with high blood pressure, although the nature, direction and implications of this association are unclear.
INTRODUCTION
Vitamin D affects various parts of the body, including the musculoskeletal, cardiovascular, and neurocognitive systems. It is also related to mental health. Low vitamin D level is associated with various psychiatric illnesses, including depression, dementia, and autism spectrum disorder [1-4]. Vitamin D levels are low in patients with schizophrenia. In one study, the prevalence of vitamin deficiency/insufficiency was 92.5% in patients in the acute phase of schizophrenia [5]. The level of vitamin D also affects symptom severity in patients with schizophrenia [6,7]. Levels of vitamin D in utero predict later risk for schizophrenia [8].
Low vitamin D level is independently associated with all-cause mortality in the general population [9]. In particular, low vitamin D is associated with cardiovascular disease [10]. In a 10-year follow-up study, male patients without previous cardiovascular disease and vitamin D deficiency [25(OH)D<15 ng/mL] exhibited a twofold increase in the rate of myocardial infarction [11]. Participants in the Framingham Offspring Study who had no history of cardiovascular disease but a severe vitamin D deficiency [25(OH)D<10 ng/mL] had a hazard ratio of 1.8 (95% confidence interval, 1.05–3.08) for developing new cardiovascular disease after a 5-year follow-up compared to participants who had higher levels of 25(OH)D (>15 ng/mL), although the direction of the association remains uncertain [10].
However, evidence demonstrating an association between vitamin D and physical health in patients with schizophrenia is scarce. Patients with schizophrenia die 10–20 years earlier than the general population [7]. This premature mortality is attributed to high comorbid physical illnesses, including cardiovascular disease and metabolic syndrome [12,13]. Thus, it is important to understand the effect of vitamin D on the physical health of patients with a psychotic disorder.
Vitamin D levels vary according to ethnicity. They were shown to be relatively high in Caucasians compared to those in Mexican-Americans and African-Americans [14]. In addition, vitamin D level also varies from region to region [15]. Asians, including Japanese and Koreans, show low vitamin D levels during summer and winter compared to people in other countries [16,17]. Thus, vitamin D levels must be studied in various regions and ethnicities. In this study, we investigated the relationship between vitamin D level and metabolic syndrome in Korean patients with psychotic disorders.
Methods
Participants
The present study used the data set from Gwangju Comprehensive Health Inspection for Mental Illness, which evaluated the physical health of patients with mental illness (psychotic and mood disorders) treated in community mental health centers in Gwangju, South Korea. The present study used data from patients who were treated for a psychotic disorder and excluded patients with a mood disorder. Of the 386 participants in the parent study, 302 with a psychotic disorder were included in the present study, of which 248 (82.1%) had schizophrenia, 36 (11.9%) had schizoaffective disorder, and 18 (6.0%) had other specified schizophrenia spectrum disorder. This study was conducted from November 2015 to January 2016 and was approved by Chonnam National University Hospital’s Institutional Review Board (CNUH-2015-254). All subjects provided written informed consent before participating.
Measures
Basic demographic and clinical information was obtained from medical records and case managers and included psychiatric diagnoses, treatment duration, prescribed medications and dosages, depressive symptoms, physical activities, and dietary habits. Risperidone-equivalent doses were calculated to assess dose-dependent drug effects [18].
The Clinical Global Impression (CGI) Scale was administered by case managers to evaluate global symptom severity [19]. Depressive symptoms were measured by four items from the Center for Epidemiological Studies Depression Scale (bothered by things, poor appetite, feel blue, and feel depressed) [20]. Physical activity was measured by three items from the International Physical Activity Questionnaire (IPAQ)-Short Form (vigorous physical activity, moderate physical activity, and walking) [21]. Physical activity was determined by the type of exercise and time according to the IPAQ scoring protocol [22]. Dietary habits were evaluated with the dietary guidelines published by the Korean Ministry of Health and Welfare [23]. The dietary questionnaire consisted of three subcategories including five items for diet regularity, six items for a balanced diet, and nine items for unhealthy diet and habits. In the balanced diet category, the dairy intake item was also used as a proxy variable for vitamin D intake. We asked how many times participants took milk and dairy products every week.
Fasting venous blood samples were obtained to assess vitamin D levels; lipid profiles including high-density-lipoprotein (HDL) cholesterol, low-density-lipoprotein cholesterol, triglycerides, and total cholesterol; fasting blood glucose (FBG); glycated hemoglobin, liver function tests including aspartate aminotransferase (AST), alanine transaminase (ALT), and gamma-glutamyltransferase; renal function tests, including blood urea nitrogen and creatinine; and prolactin. Blood pressure including systolic blood pressure and diastolic blood pressure was also measured. Body mass index was calculated as weight/height2 (kg/m2).
Metabolic syndrome was diagnosed with the definitions of the modified ATP III for Korea (three or more of the following five criteria): abdominal obesity, defined as a waist circumference ≥90 cm in men and ≥85 cm in women [24]; serum triglycerides ≥150 mg/dL (1.7 mmol/L) or drug treatment for elevated triglycerides; serum HDL cholesterol<40 mg/dL (1 mmol/L) in men and <50 mg/dL (1.3 mmol/L) in women or drug treatment for low HDL cholesterol; blood pressure ≥130/85 mm Hg or drug treatment for elevated blood pressure; and FPG ≥100 mg/dL (5.6 mmol/L) or drug treatment for elevated blood glucose.
Statistical analysis
The participants were categorized into two groups according to vitamin D level. Vitamin D insufficiency was defined as <20 ng/mL (equivalent to <50 nmol/L) and a serum level ≥20 ng/mL (equivalent to ≥50 nmol/L) was considered optimal [25].
Comparisons of demographic and clinical data according to the presence of vitamin D insufficiency were conducted with the χ2 test, independent t-test, or Mann-Whitney U-test, as appropriate. We examined the relationship between serum vitamin D insufficiency and metabolic syndrome using logistic regression analysis after controlling for physical activity and dietary habits. The statistical analysis was performed using SPSS for Windows ver. 23.0 (IBM Corp., Armonk, NY, USA). All statistical tests were two-sided, and the α-level for statistical significance was 0.05.
Results
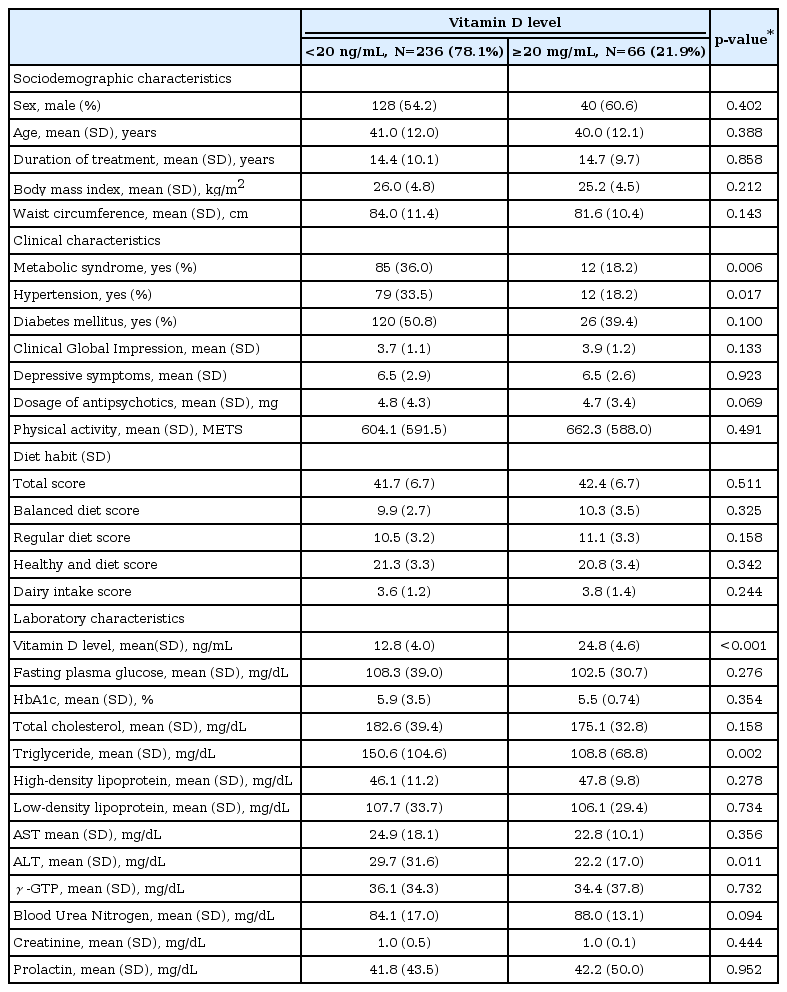
Among the 302 participants, 168 (55.6%) were males and 134 were females. Their mean age was 40.7±12.0 years (range 18–82 years) and mean duration of illness was 14.5±10.0 years. Mean vitamin D level was 15.5±6.4 ng/mL, and 236 patients (78.1%) had a vitamin D insufficiency (<20 ng/mL). Ninetyseven patients (32.1%) met the criteria for metabolic syndrome. Vitamin D level was significantly lower in patients with metabolic syndrome than in those without metabolic syndrome (13.1±5.0 ng/mL vs. 16.6±6.7 ng/mL; t=4.616, p<0.001).
Table 1 shows the demographic and clinical characteristics according to the presence of vitamin D insufficiency. Age, sex, and illness duration were not different according to vitamin D insufficiency. Metabolic syndrome and hypertension were significantly more frequent and levels of triglycerides and ALT were significantly higher in patients with a vitamin D insufficiency than in those without a vitamin D insufficiency. Values of other laboratory measures were not different based on the presence of a vitamin D insufficiency. Scores on the CGI, the depression scale, IPAQ, and the dietary questionnaire were also not different according to vitamin D insufficiency.
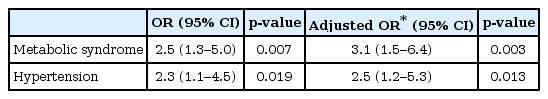
Table 2 shows the results of the logistic regression analysis. Vitamin D insufficiency was significantly associated with metabolic syndrome and hypertension. After adjusting for physical activity and dietary habit scores, vitamin D insufficiency remained significantly associated with metabolic syndrome and hypertension.
Discussion
Vitamin D deficiency is considered a major health issue worldwide, although its significance is uncertain [17]. This also applies to the psychiatric field [3]. In general medicine, the link between Vitamin D and medical comorbidity has shifted from being seen as causative to being a proxy marker of lifestyle issues, highlighted by the failure of many treatment trials to influence the targeted disorder [26]. The present study demonstrated that vitamin D insufficiency was highly prevalent (78.1%) in patients with a psychotic disorder and was associated with metabolic syndrome and hypertension. The high prevalence of vitamin D insufficiency in our population with a psychotic disorder was compatible with previous studies conducted in western countries [3,27]. The prevalence of vitamin D insufficiency in this community-dwelling psychotic population was higher than that in the general South Korean population. A recent South Korean study of 17,252 health examinees showed that 60.4% (47.3% in males and 64.5% in females) of examinees had a vitamin D insufficiency (<20 ng/mL) [28].
The prevalence of metabolic syndrome in this study was 32.1%, which is higher than that in the South Korean general population [29,30]. We also observed that metabolic syndrome components, such as hypertension and high triglyceride, were inversely associated with circulating serum vitamin D levels. Metabolic syndrome is a very important risk factor for cardiovascular disease, which is a major cause of death and early mortality in patients with psychosis [12,31,32]. In previous studies of the general population, vitamin D insufficiency was associated with a risk for metabolic syndrome [33-35]. Vitamin D deficiency is also related to increased risk of hypertension, heart failure, and ischemic disease [10]. This is the first study to demonstrate an association between vitamin D insufficiency, metabolic syndrome, and hypertension in an Asian population with psychosis.
Several mechanisms have been proposed to explain the association between vitamin D and future risk of metabolic syndrome. Since vitamin D is fat soluble and could be stored in adipose tissue, the levels of circulating vitamin D can be reduced in the blood of obese individuals [35,36]. Vitamin D has also been shown to inhibit the release of cytokines from the immune cell [37]. Another hypothesis is the negative regulatory effect of vitamin D on renin activity. In an animal study, vitamin D receptor knockout mice had hypertension, cardiac hypertrophy, and increased activation of the renin-angiotensin-aldosterone system [38]. Mechanism for the association between vitamin D and cardiovascular risk involves the effect of vitamin D on improving insulin sensitivity [39,40] by directly acting on cardiac tissue in response to injury [41] and vascular compliance and response to injury [42,43].
As this study was cross-sectional, we cannot conclude that vitamin D insufficiency was the cause of metabolic syndrome or hypertension. In particular, poor physical activity and dietary habits may affect both metabolic syndrome and vitamin D insufficiency because vitamin D is produced in the skin as vitamin D3 through sun exposure or is obtained from the diet as vitamin D2 or D3 [10]. In our study, only 66 patients (21.9%) were moderately or highly physically active as measured by the IPAQ. This is a very low level compared with levels of physical activity measured by the same scale for the general populations in 20 other countries [44]. Negative symptoms of schizophrenia may be related to low physical activity [45] and sun exposure, and consequently cause a vitamin D insufficiency. However, in our study, vitamin D level was not different according to physical activity or dietary habits. In addition, the significant association between vitamin D insufficiency and metabolic syndrome remained after adjusting for physical activity and dietary habits.
Although liver function tests including AST and ALT are not diagnostic criteria for metabolic syndrome, liver markers have also been implicated in the development of metabolic syndrome [46]. ALT is correlated with increased hepatic fat content, which is associated with insulin resistance regardless of body weight. Insulin resistance has detrimental effects on components of metabolic syndrome [47-49]. Our study showed that patients with a vitamin D insufficiency were associated with a higher ALT, which indirectly suggests an association between vitamin D insufficiency and metabolic syndrome.
Whether vitamin D supplementation decreases cardiovascular risk in the general population or patients with mental illness is controversial. Several studies have focused on vitamin D as a potential anti-hypertensive agent, but they have not consistently shown a positive effect [10]. Very few studies have been performed on whether vitamin D supplementation reduces the risk of cardiovascular mortality. Both the Women’s Health Initiative Study [50] and a European study [51] showed no improved cardiovascular survival after vitamin D supplementation. On the other hand, a recent meta-analysis examined nine randomized controlled trials and showed an 8% reduction in all-cause mortality with supplementation of up to 500 IU vitamin D [10]. That study was conducted on a general population. Although a few studies have analyzed whether vitamin D supplementation affects mental health, with the largest being negative [52], no research has been conducted on the influence of vitamin D supplementation on metabolic syndrome and cardiovascular risk in patients with a psychotic disorder. In addition, there is no consensus on the appropriate vitamin D dose for patients with a mental illness. Therefore, prospective studies are required to define whether vitamin D supplementation can reduce the occurrence of metabolic syndrome and cardiovascular risk in patients with a psychotic disorder and the appropriate vitamin D dose needed.
Vitamin D level is associated with various mental health issues. In particular, low vitamin D level is associated with depression [53]. However, in our study, low vitamin D level was not related to depressive symptoms or the general severity of psychotic symptoms.
This study had several limitations. First, this study was crosssectional, so we were unable to investigate the causal relationship between vitamin D and metabolic syndrome. Second, the assessment of depressive symptoms, dietary habits, and physical activity depended on self-reported data. Third, a formal scale to measure psychotic symptoms was not administerd. Fourth, we did not investigate intake of vitamin supplements. Finally, most of the study population participated in this study during late fall and early winter when vitamin D levels might be lower than at other times of the year [28,54]. In addition, amount of sun exposure was not investigated.
Nevertheless, this study has important clinical and research implications by showing that vitamin D insufficiency was very prevalent and associated with metabolic syndrome and hypertension in South Korean patients with psychosis. Further longitudinal and interventional studies are required in patients with psychosis.
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2014R1A1A4A01005245). This research work was conducted as part of the Gwangju Mental Health Demonstration Project supported by Ministry of Health & Welfare and Gwangju Metropolitan city, Republic of Korea.