|
 |
- Search
| Psychiatry Investig > Volume 15(2); 2018 > Article |
|
Abstract
Objective
Stroke is associated with significant long-term morbidity and poor quality of life (QOL). Depression is one of the most common complications after stroke and has been associated with QOL cross-sectionally. We investigated the longitudinal impact of depression in the acute phase of stroke on QOL 1 year after stroke.
Methods
In total, 423 patients were evaluated 2 weeks after stroke, and 288 (68%) were followed 1 year later. QOL was assessed using the World Health Organization Quality of Life-Abbreviated form (WHOQOL-BREF) at baseline and follow-up. Depression was diagnosed according to Diagnostic and Statistical Manual of Mental Disorders-IV criteria; demographic and clinical characteristics data, including stroke severity, were obtained at baseline. The longitudinal associations of post-stroke depression (PSD) at baseline with QOL across two evaluation points were assessed using a repeated-measures analysis of variance.
Stroke is a leading cause of disability worldwide [1]. According to the World Health Organization, 15 million people suffer a stroke worldwide each year, and, of those, 5 million die and another 5 million are permanently disabled. Despite significant therapeutic advances in post-stroke management in the acute phase, the application of secondary prevention principles in clinical practice is far from optimal [2]. Accordingly, quality of life (QOL) and psychological well-being are important targets for secondary prevention in post-stroke patients [3]. Therefore, after intensive medical care in the acute phase of stroke, QOL is increasingly considered a relevant outcome measure in stroke survivors with relatively stable symptoms.
The determinants of QOL in stroke survivors have been widely investigated. Several physical, social, functional, and psychological factors, including age, sex, socioeconomic status, the presence of comorbid conditions, level of education, physical functioning, stroke severity, cognition, and depression, have been identified as key as determinants of QOL [4-7]. Depression, in particular, has significant adverse effects on QOL in stroke patients [8]. A systematic review and meta-analysis using pooled data from 43 cohorts and more than 20,000 patients conducted in 2013 found that depression was common in stroke patients, with a prevalence of 29% (95% confidence interval, 25-32%) observed any time after stroke [9].
Most previous studies have investigated the cross-sectional associations between depression and QOL at various time points after stroke. In an Italian multicenter observational study (DESTRO), Paolucci et al. [10] found that post-stroke depression (PSD) was correlated with lower QOL in the acute phase of stroke (1 month after the index stroke). In a study of patients with intracerebral hemorrhage (ICH) in the subacute phase of stroke (1-3 months after the index stroke), Christensen et al. [11] found that the severity of depressed mood at day 90 post-stroke was significantly correlated with poor QOL irrespective of post-ICH disability and impairment. These findings suggest that PSD affects QOL in the acute/subacute phase despite physical medical treatment. Pan et al. [8] investigated patients in the chronic phase of stroke (3 months after the index stroke) and found that health-related QOL domain scores were primarily associated with depression 3, 6, and 12 months after stroke. Additionally, Naess et al. [12] reported that fatigue and depression were major independent variables correlated with low QOL after a mean follow-up period of 6 years. Thus, the findings of previous studies indicate that depression is a major independent variable that is cross-sectionally correlated with low QOL at any time after stroke. However, cross-sectional associations cannot explain cause and effect relationships between PSD and QOL.
To our knowledge, no previous longitudinal studies have investigated whether PSD can predict QOL. Therefore, we used data from a longitudinal study of a post-stroke cohort to investigate the associations between PSD and QOL at 2 weeks and 1 year after stroke to determine whether PSD at baseline predicted poor QOL 1 year after a stroke.
Our study is a component of a larger parent study of neurological and psychiatric morbidity in stroke survivors using a naturalistic prospective design, which has been described in detail previously [13]. In brief, participants were consecutively recruited from all recent ischemic stroke patients hospitalized in the Department of Neurology of Chonnam National University Hospital in Gwangju, South Korea. Assessments were made at 2 weeks (baseline) and at 1 year (follow-up) after stroke to investigate the acute and chronic consequences of the event. PSD and demographic and clinical covariates were evaluated at 2 weeks, and QOL was assessed 2 weeks and 1 year after stroke.
At about 2 weeks post-stroke, all potentially eligible patients were approached about participation in the study. The inclusion criteria were: 1) ischemic stroke confirmed by brain magnetic resonance imaging (MRI) or computed tomography (CT; if MRI was contraindicated); 2) ability to complete the necessary investigations and questionnaires; and 3) capacity to understand the objective of the study and provide informed consent. The exclusion criteria were: 1) severe physical illnesses that were life-threatening or interfered with the recovery from stroke; 2) communication difficulties due to dysphasia or dysarthria precluding informed consent and completion of the questionnaires; 3) any of the following comorbid neuropsychiatric conditions: dementia, Parkinson’s disease, brain tumor, epilepsy, psychoses, and alcohol and substance dependence; 4) severe physical illnesses limiting movement prior to stroke; and 5) a Mini-Mental State Examination (MMSE) score of <16 [14]. Patients were recruited for the initial 2-week assessment between 2006 and 2010 and attempts were made to follow up all participants after 1 year. All participants provided written informed consent. This study was approved by the Chonnam National University Hospital Institutional Review Board (IRB No. I-2008-02-028).
QOL was measured using the World Health Organization Quality of Life-Abbreviated form (WHOQOL-BREF), a 26-item self-administered questionnaire in which items are rated on a 5-point scale. The WHOQOL-BREF evaluates domains related to physical factors, psychological factors, social relationships, and environmental context. Raw subscale scores are converted to a 0-100 scale to facilitate comparisons with other data sets, with higher scores indicating a better QOL. The WHOQOL-BREF was formerly translated and standardized in Korean [15].
Depressive disorders after stroke were diagnosed according to Diagnostic and Statistical Manual of Mental Disorders-IV (DSM-IV) [16] criteria using the Mini International Neuropsychiatric Interview (MINI), a structured psychiatric interview for DSM-IV depression categories. According to the criteria, patients were diagnosed as having depression if they had at least one core symptom (i.e., depressed mood or loss of interest) and at least two other symptoms of depression. The MINI has been formally translated and standardized in Korean [17].
Characteristics potentially associated with stroke outcomes were considered as covariates in our analysis. Data on age, sex, years of education, and previous histories of depression and stroke were obtained. Stroke hemisphere and location were assessed using brain MRI or CT images. Stroke severity was assessed using the National Institutes of Health Stroke Scale (NIHSS; scores range from 0-42, with higher scores indicating more severe pathology) [18], physical disability was measured using the Barthel Index (BI; scores range from 0-100, with lower scores indicating more severe disability) [19], and cognitive function was evaluated using the MMSE (scores range from 0-30, with lower scores indicating lower cognitive function) [14]. Scores on the NIHSS, BI, and MMSE were obtained at admission. All tests were administered by a trained interviewer.
Patients were divided into groups according to whether PSD was present or absent at baseline. Demographic and clinical characteristics were compared between groups using t-tests, chi-squared tests, or Fisher’s exact tests as appropriate. Those variables showing significant associations with PSD (p-value <0.05) were used as covariates in multivariate analyses. The cross-sectional associations between PSD and QOL were evaluated using t-tests for the unadjusted model and then using logistic regression tests after adjustment for those demographic and clinical characteristics significantly associated with PSD. A repeated-measures analysis of variance (ANOVA) using the same adjustment model was performed to assess the longitudinal associations of PSD at baseline with WHOQOL-BREF scores across evaluation points. Statistical analyses were conducted using the Statistical Package for the Social Sciences ver. 23.0 (IBM Corp., Armonk, NY, USA).
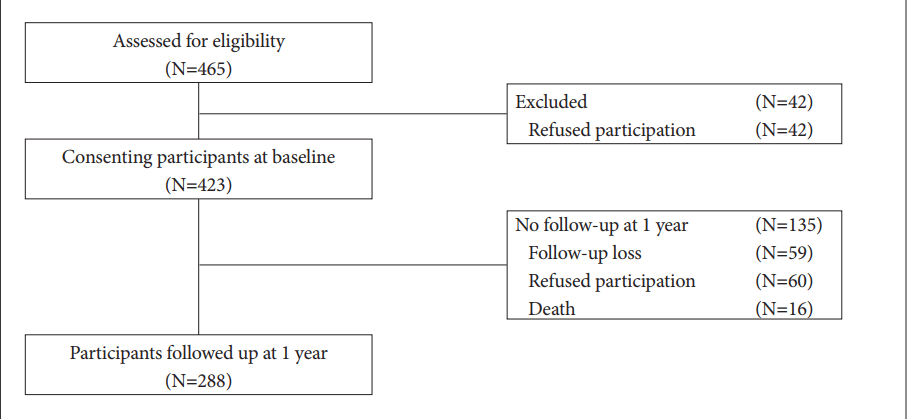
The recruitment process is shown in Figure 1. Of 465 consecutively enrolled potentially eligible stroke patients, 423 (91%) consented to participate in the study. No significant differences were found between participants and non-participants with respect to any demographic or clinical characteristics (all p-values >0.1). In total, 288 (70%) patients were re-examined after 1 year. The baseline demographic and clinical characteristics of patients who were followed up did not differ significantly from those who were not followed up (all p-values >0.1). The mean (standard deviation) time from admission to 2-week assessment was 12.3 (3.0) days, and the time from admission to 1-year follow up was 13.2 (3.6) months.
Of the patients included in the analysis (n=423), PSD was present in 103 (24.3%) at baseline. The mean (SD) age was 64.5 (10.0; range, 30-87) years, 244 (57.7%) were male, and the mean (SD) duration of education was 8.5 (5.0) years. Overall, 17 participants reported previous depression (4.0%) and 35 participants reported a previous stroke (8.3%). The mean (SD) score on the NIHSS was 3.5 (3.2), while that on the BI was 80.1 (22.9). Stroke laterality was left hemisphere in 187 (44.2%), right hemisphere in 216 (51.1%), and bilateral in 20 (4.7%) patients. These characteristics are shown according to PSD status at baseline in Table 1. Compared with patients without PSD, those with PSD were significantly older, more likely to have a history of depression and/or stroke, more likely to have had a stroke involving the anterior circulation, and, as a group, had higher NIHSS scores and lower BI and MMSE scores.
The cross-sectional associations between PSD and QOL at baseline are shown in Table 2. PSD was significantly associated with all four domains of the WHOQOL-BREF before and after adjustment for age, history of depression and stroke, stroke location, and scores on the NIHSS, BI, and MMSE.
Of the 288 patients evaluated at follow up, 66 (22.9%) had PSD at baseline. The associations of PSD at baseline with the four WHOQOL-BREF domains (physical health, psychological health, social relationships, and environmental context) at baseline and follow up are shown in Figure 2. A repeated-measures ANOVA revealed a significant effect of PSD group on the four WHOQOL-BREF domain scores after adjustment for age, history of depression and stroke, stroke location, and NIHSS, BI, and MMSE scores. However, no significant PSD group by time interaction was found for WHOQOL-BREF scores, indicating that the WHOQOL-BRED scores were significantly and persistently lower in patients with PSD than in those without PSD.
The major finding of our longitudinal study of a post-stroke cohort is that PSD had a significant and persistent negative impact on QOL at 2 weeks and 1 year after stroke. The negative effect of PSD was evident in the four WHOQOL-BREF domains and remained significant after adjusting for relevant covariates. These findings suggest that PSD in the acute phase of stroke is an independent predictor of various aspects of QOL in the acute and chronic phases of stroke regardless of demographic and clinical characteristics, including stroke severity.
Our finding that PSD is associated with low QOL in the acute phase of stroke is consistent with previous findings that post-stroke depressive symptoms are associated with poor post-stroke QOL [11,20-22], suggesting that PSD is closely related to QOL. Although medical treatment is given priority in the management of stroke patients during the acute phase and psychological considerations become more important during the rehabilitation or chronic phases of stroke, our findings highlight the importance of the early evaluation and management of PSD.
The main strength of our study, and the factor that distinguishes it from previous studies, is that we evaluated the long-term effects of PSD on QOL in patients diagnosed with PSD in the acute phase. To our knowledge, the longitudinal association between PSD and QOL have not been assessed in a meta-analysis previously [23], and we are the first to report a significant relationship between PSD in the acute phase of stroke and multi-domain QOL scores 1 year after stroke. Previous studies have shown an association between depression and poor QOL in the acute/subacute phases of stroke [10,11], and in the chronic phase determined cross-sectionally [12,22]. We found that PSD present in the acute phase predicted low QOL persisting from the acute to the chronic phase of stroke. Moreover, these associations were independent of stroke severity. Our findings highlight the negative impact of depression after stroke and suggest the need for the mandatory assessment of depression in the acute phase of stroke.
The QOL of patients with baseline PSD might be expected to improve or worsen over time as stroke symptoms stabilize. However, although the effect of group was significant, we did not find a significant interaction between time and group (Figure 2). Thus, our findings suggest that the presence of PSD in the acute phase of stroke has persistent, modifying negative effects on the trajectory of QOL from the acute to chronic phase of stroke.
Another strength of our study is that it excluded proxy bias. Physical, cognitive, and language impairments in stroke survivors may necessitate proxy responses for self-reported outcomes. A previous study found that proxies tended to report worse QOL scores than did stroke survivors themselves [24]. In our study, QOL was measured using the WHOQOL-BREF self-report questionnaire, which has the advantage of being able to exclude proxy bias. A third strength of our study is that depression and clinical covariates were assessed at similar time points (2 weeks after stroke) in all participants. The etiology of depression has been reported to differ according to the time elapsed after stroke, with biological factors more important at the acute phase and psychosocial factors increasing in later on [25]. Therefore, we focused on the acute post-stroke period to reduce the risk of bias arising from assessments at varying time points from stroke onset. We used a structured diagnostic interview based on DSM-IV criteria to diagnose depression and found PSD in 24.3% of the stroke patients. This prevalence is comparable to, although slightly lower than, findings from previous studies using a similar design, diagnostic criteria, and time points since stroke [26]. An additional advantage of our study is that the participants were consecutive recent stroke patients from the same institution (our hospital), which reduced the likelihood of selection bias and increased the potential generalizability.
Our study has several limitations. We did not treat PSD and we did not assess PSD at the 1-year follow up. However, our findings suggest that the presence of PSD in the acute phase of stroke was associated with lower QOL in the chronic phase regardless of treatment. Further examination of the associations between PSD and QOL in relation to the time course and treatment response of depression is needed. Furthermore, because we excluded patients with severe cognitive impairments or aphasia who were unable to complete the self-rating scales or psychiatric interview, our findings must be interpreted with caution as they may only apply to patients with mild-to-moderate stroke severity without cognitive and language deficits. However, as noted above, excluding such patients allowed us to rule out proxy biases.
In conclusion, the findings of our 1-year longitudinal study showed that acute phase PSD had a negative impact on QOL in both the acute and chronic phases of stroke after adjusting for stroke severity, physical disability, and cognitive function. Our findings underscore the importance of diagnosing and evaluating PSD during the acute phase of stroke to predict long-term QOL. The fact that PSD is underdiagnosed and undertreated is well documented [27]. The challenge is to raise awareness of this issue and promote the assessment of depression in the acute phase of stroke. Strategies to manage PSD, in addition to medical treatment, must be developed to improve the QOL of stroke survivors.
Moreover, further research is needed to determine whether PSD treatment can improve QOL and to develop strategies to improve QOL in individuals with or at risk of developing PSD. Antidepressant drugs may play a relevant role in improving depressive symptoms and reducing the negative impact of PSD [28]. Finally, because stroke survivors are living longer, it is necessary to confirm our findings in longer-term studies.
ACKNOWLEDGEMENTS
This study was supported by a grant of National Research Foundation of Korea Grant (NRF-2015M3C7A1028899) to Prof. JM Kim.
Figure 2.
Longitudinal associations between each four domains of WHOQOLBREF(physical health; psychological health; social relationships; environmental context) scores and PSD status adjusted for all covariants.

Table 1.
Baseline sample characteristics by post-stroke depression (PSD) status
Table 2.
Cross-sectional associations of post-stroke depression (PSD) and quality of life at baseline
REFERENCES
1. Roger VL, Go AS, Lloyd-Jones DM, Adams RJ, Berry JD, Brown TM, et al. Heart disease and stroke statistics--2011 update: a report from the American Heart Association. Circulation 2011;123:e18-e209.


2. Heuschmann PU, Kircher J, Nowe T, Dittrich R, Reiner Z, Cifkova R, et al. Control of main risk factors after ischaemic stroke across Europe: data from the stroke-specific module of the EUROASPIRE III survey. Eur J Prev Cardiol 2015;22:1354-1362.


3. Kielbergerová L, Mayer O Jr, Vaněk J, Bruthans J, Wohlfahrt P, Cífková R. Quality of life predictors in chronic stable post-stroke patients and prognostic value of SF-36 score as a mortality surrogate. Transl Stroke Res 2015;6:375-383.


4. Niemi ML, Laaksonen R, Kotila M, Waltimo O. Quality of life 4 years after stroke. Stroke 1988;19:1101-1107.


5. De Haan RJ, Limburg M, Van der Meulen JH, Jacobs HM, Aaronson NK. Quality of life after stroke. Stroke 1995;26:402-408.


6. Carod-Artal J, Egido JA, González JL, de Seijas EV. Quality of life among stroke survivors evaluated 1 year after stroke: experience of a stroke unit. Stroke 2000;31:2995-3000.


7. Williams LS, Kroenke K, Weinberger M, Harris LE, Biller J. Post-stroke depression affects quality of life (QOL) but not other stroke outcome measures. Stroke 1999;30:236-239.
8. Pan JH, Song XY, Lee SY, Kwok T. Longitudinal analysis of quality of life for stroke survivors using latent curve models. Stroke 2008;39:2795-2802.


9. Ayerbe L, Ayis S, Wolfe CD, Rudd AG. Natural history, predictors and outcomes of depression after stroke: systematic review and meta-analysis. Br J Psychiatry 2013;202:14-21.


10. Paolucci S, Gandolfo C, Provinciali L, Torta R, Toso V, DESTRO Study Group. The Italian multicenter observational study on post-stroke depression (DESTRO). J Neurol 2006;253:556-562.


11. Christensen MC, Mayer SA, Ferran JM, Kissela B. Depressed mood after intracerebral hemorrhage: the FAST trial. Cerebrovasc Dis 2009;27:353-360.


12. Naess H, Waje-Andreassen U, Thomassen L, Nyland H, Myhr KM. Health-related quality of life among young adults with ischemic stroke on long-term follow-up. Stroke 2006;37:1232-1236.


13. Kim JM, Stewart R, Bae KY, Kim SW, Kang HJ, Shin IS, et al. Serotonergic and BDNF genes and risk of depression after stroke. J Affect Disord 2012;136:833-840.


14. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc 1997;15:300-308.
15. Min SK, Lee CI, Kim KI, Suh SY, Kim DK. Development of Korean version of WHO quality of life scale abbreviated version (WHOQOL-BREF). J Korean Neuropsychiatr Assoc 2000;39:571-579.
16. Sheehan D, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. Diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 1998;59(Suppl 20):22-33.
17. Yoo SW, Kim YS, Noh JS, Oh KS, Kim CH, NamKoong K, et al. Validity of Korean version of the mini-international neuropsychiatric interview. Anxiety Mood 2006;2:50-55.
18. Kasner SE, Chalela JA, Luciano JM, Cucchiara BL, Raps EC, McGarvey ML, et al. Reliability and validity of estimating the NIH stroke scale score from medical records. Stroke 1999;30:1534-1537.


19. Mahoney FI, Barthel DW. Functional Evaluation: the Barthel Index. Md State Med J 1965;14:61-65.
20. Naess H, Waje-Andreassen U, Thomassen L, Nyland H, Myhr KM. Health-related quality of life among young adults with ischemic stroke on long-term follow-up. Stroke 2006;37:1232-1236.


21. Gandolfo C, Provinciali L, Torta R, Toso V, DESTRO Study Group. The Italian multicenter observational study on post-stroke depression (DESTRO). J Neurol 2006;253:556-562.


22. Sturm JW, Donnan GA, Dewey HM, Macdonell RA, Gilligan AK, Srikanth V, et al. Quality of life after stroke: the North East Melbourne Stroke Incidence Study (NEMESIS). Stroke 2004;35:2340-2345.


23. Towfighi A, Ovbiagele B, El Husseini N, Hackett ML, Jorge RE, Kissela BM, et al. Poststroke depression: a scientific statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2017;48:e30-e43.


24. Williams LS, Bakas T, Brizendine E, Plue L, Tu W, Hendrie H, et al. How valid are family proxy assessments of stroke patients’ health-related quality of life? Stroke 2006;37:2081-2085.


25. Bhogal SK, Teasell R, Foley N, Speechley M. Lesion location and poststroke depression: systematic review of the methodological limitations in the literature. Stroke 2004;35:794-802.


26. Berg A, Palomäki H, Lehtihalmes M, Lönnqvist J, Kaste M. Poststroke depression in acute phase after stroke. Cerebrovasc Dis 2001;12:14-20.