196G/A of the Brain-Derived Neurotrophic Factor Gene Polymorphisms Predicts Suicidal Behavior in Schizophrenia Patients
Article information
Abstract
Objective
Brain-derived neurotrophic factor (BDNF) has possible neurobiologic impact on etiology of schizophrenia. We hypothesized that the specific allele or the genotype such as two single nucleotide polymorphisms (SNPs) , 196G/A (rs6265), 11757G/C(rs16917204) is associated with schizophrenia or its clinical features.
Methods
241 normal controls and 157 schizophrenia patients are included. The differences in allele or genotype distribution for the patients and normal controls were analyzed. We also analyzed clinical variables among patients.
Results
We found no significant difference in genotype or allele distributions of two studied SNPs between the patient group and the control group. However, history of suicide attempt was relatively higher in patients with genotype with A allele, compared to patients with genotype G/G for 196G/A (p-value=0.045).
Conclusion
Our results suggest that it is possible to use BDNF gene allele and genotype as a predictor for suicide attempt in schizophrenia patients. It can help manage the schizophrenia patients regarding suicidal behavior and furthermore, mortality.
INTRODUCTION
Schizophrenia is a psychiatric disorder characterized by delusions, hallucinations, disorganized behaviors and languages [1]. Treatment response has increased with the use of typical and atypical antipsychotics. However, approximately 20% to 30% of patients do not respond to medication and are considered treatment-resistant patients [2,3]. Understanding the genetics and neurobiological basis of schizophrenia and characteristics of each patients is necessary for improved treatment responses.
Researchers have attempted to discover the genetic basis of psychiatric disorders, including schizophrenia. Brain-derived neurotrophic factor (BDNF) is a highly expressed neurotrophin in the prefrontal cortex and hippocampus [4]. BDNF has long-term effects on neuronal survival, differentiation, synaptic plasticity [5], development of the central nervous system (CNS) in children and maintenance of the CNS in adults [6].
Whether BDNF is associated with schizophrenia has been investigated [7] and many models considering such association have been proposed. BDNF modulates the expression of dopamine D3 receptors [8] and contributes to maturation and plasticity of dopaminergic pathways, including the mesolimbic pathway [9]. BDNF modulates the firing rates of serotonergic neurons within the raphe nuclei [10] and this relationship with serotonergic signaling contributes to schizophrenia.
Different effects of single nucleotide polymorphism (SNP) on BDNF have been studied. One of the most studied SNPs, 196G/A (Val66Met, rs6265), plays a modulating role in BDNF activity. SNP 196G/A also showed an association with cognitive impairment and brain volume differences in human studies [11,12]. Another well-studied SNP, 11757G/C (rs16917204), was associated with Alzheimer’s disease with comorbid depression [13,14]. Many studies from different groups proposed SNP 11757G/C was associated with various psychiatric disorders, although consensus has not been reached.
Aims of the study
We hypothesized specific BDNF gene polymorphisms contribute to susceptibility to schizophrenia. We also hypothesized specific BDNF gene allele and genotype may have relationship with specific clinical characteristics of each patients. Discovering any associations will help the understanding of the neurobiological background of schizophrenia and provide newer treatment options.
METHODS
A total of 157 patients diagnosed with schizophrenia based on the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) were enrolled in the present study. We recruited patients who admitted Korea University Ansan Hospital due to acute psychotic symptoms from 2006 to 2011. Each patient received a Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I) [15] performed by trained psychiatrists. Subjects with comorbid psychiatric disorders, such as mood disorder, anxiety disorder, neurocognitive disorder or schizoaffective disorder were excluded.
Patient clinical data and past personal history were assessed. Demographic data including age at onset, number of admissions and disease duration, history of suicide attempts were collected. For clinical assessment, severity of disease was evaluated by trained psychiatrists using the Positive And Negative Syndrome Scale (PANSS) [16], Brief Psychiatric Rating Scale (BPRS) [17], and Korean version of the Calgary Depression Scale for Schizophrenia (K-CDSS) [18].
The normal control group included 241 healthy individuals who visited Korea University Ansan Hospital for regular health checkups. Subjects with a history of personal or familial psychiatric diagnoses, or who had a Beck Depression Inventory (BDI) score higher than 10 [19] or State-Trait Anxiety Inventory (STAI) higher than 40 were excluded [20]. Written informed consent was obtained from all subjects. The study protocol was approved by the Ethics Committee of Korea University Ansan Hospital (2005AS0008).
DNA analyses and genotyping were performed for both patients and controls. We examined 2 BDNF SNPs, 196G/A (rs6265), and 11757G/C (rs16917204). The genotyping process was performed as follows:
For 196A site, DNA was extracted from blood leukocytes using a commercial DNA extract kit, Wizard Genomic DNA purification kit (Promega, WI, USA). Polymerase chain reaction (PCR) was performed with the forward primer 5'-GAG GCT TGA CAT CAT TGG CT-3' and the reverse primer 5'-CGT GTA CAA GTC TGC GTC CT-3'. The amplification mixture contained 0.5 uL of 100 ng/uL DNA, 2.5 uL of 10× Taq buffer, 0.5 uL of 10 mM dNTP mixture, 1 uL primers, 19.375 uL distilled water and 0.125 uL Taq DNA polymerase (SolGent, Seoul, Korea).
Samples were amplified using a Thermocycler (Veriti 96-well thermal cycler, Applied Biosystems) for 35 cycles. After an initial 10 min at 95°C, each cycle consisted of 30 sec at 94°C, 30 sec at 62°C and 30 sec at 72°C. After a final 5 min at 72°C, the reaction was terminated at 4°C. The amplified DNA was digested with the restriction enzyme NIaIII (New England Biolabs, MA, USA), which cuts at the 196A site, and the product was electrophoresed in 3% agarose gels and stained with ethidium bromide. Homozygous genotypes were identified based on the presence of 113 bp bands (G/G) or 75 and 38 bp bands (A/A). The heterozygous genotype had the following 3 bands: 113, 75 and 38 bp (G/A). PCR was performed with the forward primer 5'-CCT CCT GCA GCC ATT AGT-3' and the reverse primer 5'-AAT ACA AGT AGG ACC CTA GC-3'. The amplification mixture contained 0.5 uL of 100 ng/uL DNA, 2.5 uL of 10× Taq buffer, 0.5 uL of 10 mM dNTP mixture, 1 uL primers, 19.375 uL distilled water and 0.125 uL Taq DNA polymerase (SolGent).
For 11757C site, PCR was performed with the forward primer 5'-CCT CCT GCA GCC ATT AGT-3' and the reverse primer 5'-AAT ACA AGT AGG ACC CTA GC-3'. The amplification mixture contained 0.5 uL of 100 ng/uL DNA, 2.5 uL of 10× Taq buffer, 0.5 uL of 10 mM dNTP mixture, 1 uL primers, 19.375 uL distilled water, and 0.125 uL Taq DNA polymerase (SolGent).
Samples were amplified using a Thermocycler (Veriti 96-well thermal cycler, Applied Biosystems) for 35 cycles. After an initial 10 min at 95°C, each cycle consisted of 30 sec at 94°C, 30 sec at 58°C and 30 sec at 72°C. After a final 5 min at 72°C, the reaction was terminated at 4°C. The amplified DNA was digested with the restriction enzyme AvaII (New England Biolabs), which cuts at the 11757C site, and the product was electrophoresed in 3.5% agarose gels and stained with ethidium bromide. Homozygous genotypes were identified based on the presence of 65, 61, and 44 bp bands (C/C) or 105 and 65 bp bands (G/G). The heterozygous genotype had the following 4 bands: 105, 65, 61, and 44 bp (C/G).
The demographic data were compared between patient and control groups using χ2 and independent t-tests. For each SNP, whether genotype distribution followed the Hardy-Weinberg equilibrium was tested using the χ2 test. The genotype and allele frequencies of patients and controls were analyzed using χ2 tests. One-way ANOVA was used for comparison of each SNP genotype and clinical scale, clinical variables. Statistical analysis was performed using SPSS version 21.0 for Windows (IBM Corp., Armonk, NY, USA). Statistical significance level was set at p<0.05 for all statistical analyses.
RESULTS
We compared demographic data between schizophrenia patients (68 males and 89 females) and healthy controls (121 males and 120 females). The mean age in the patient group was 33.2 years with a standard deviation (SD) of 10.0 years and mean age in the control group was 31.7 years with an SD of 8.4 years. There were no significant gender or age distribution differences between the patient and control groups.
In patients with schizophrenia, the mean age at disease onset was 27.3 years (SD, 9.8 years) and mean number of admissions was 1.3 (SD, 1.9). Mean duration of disease was 72.1 months (SD, 77.3 months).
The distribution of BDNF 196G/A (rs6265), and 11757G/C (rs16917204) polymorphisms followed the Hardy-Weinberg equilibrium. The Hardy-Weinberg equilibrium values for schizophrenia patients for BDNF 196G/A were χ2=3.1408, df=1, p=0.07 and of controls χ2=0.3678, df=1, p=0.5442. For BDNF 11757G/C, the equilibrium values for schizophrenia patients were χ2=0.0534, df=1, p=0.8172 and of controls χ2=2.0874, df=1, p=0.1485.
The genotype and allele frequencies for the 2 SNPs were not different between patients and controls (Table 1).
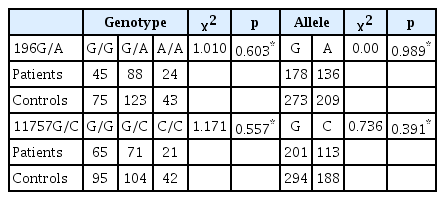
Genotype and allele frequencies of BDNF SNPs 196G/A, and 11757G/C in schizophrenia patients and normal controls
We studied the clinical variables, genotype and allele distributions among schizophrenia patients. Associations between the assessed scales and genotype distribution were not found. For 196G/A, 3 different genotypes had a mean total PANSS score ranging from 96.455 to 97.268, showing no group differences. For further evaluation, we divided total PANSS score by subscales, positive, negative, general psychopathology each. For 196G/A, mean positive score for each genotype ranged from 24.391 to 25.929. Mean negative subscale score ranged from 22.434 to 23.391, and mean general psychopathology subscale ranged from 48.171 to 48.268. For 11757G/C, no group difference was found among positive, negative, general psychopathology subscales. All those assessed scales and genotype distribution did not show any differences among genotype groups. For both K-CDSS and BPRS, no difference was found among each genotype of 196G/A. For 11757G/C genotype groups, both K-CDSS and BPRS showed no group differences.
Another clinical variable, number of admission and total duration of disease were also assessed. For 196G/A and 11757G/C, mean number of admission ranged from 0.593 to 1.477 and 1.265 to 1.286, each. For mean total duration of disease, it ranged from 39.235 to 88.309 months and from 51.0 to 77.172 months for 196G/A and 11757G/C, each. There was no group difference for both number of admission and total duration of disease.
There were no BDNF 196G/A or 11757G/C allele or genotype differences between subjects with or without a previous suicide attempt, with the following exception: there was a higher frequency of suicide attempts among patients carrying the 196G/A A allele (196G/A heterozygotes and 196 A/A homozygotes), than carriers of the 196 G/G genotype (Table 2).
DISCUSSION
Many researchers have attempted to determine correlations between psychiatric disorders and BDNF SNP genotypes; however, results among different disorders were inconsistent. A Scottish study enrolled 320 probands diagnosed with schizophrenia and 350 normal controls. Allele G of 196G/A showed strong association with schizophrenia but not bipolar disorder, compared to normal controls [21]. Conversely, a Asian study showed negative results; the G/G, G/A and A/A genotype distribution of 196G/A was not different between schizophrenia patient group and healthy control groups (p=0.31) [22]. Regarding 11757G/C, no allele or genotype difference was found when comparing schizophrenia patients with normal controls [7].
In this study, we compared genotype distribution differences for 2 SNPs, 196G/A and 11757G/C between schizophrenia patients and controls. For genotype and allele distributions, no significant association with schizophrenia was found. Our results were in agreement with the Dutch study and other previous studies [7,23,24]. The results were consistent among the studies indicating BDNF SNP genotyping alone cannot predict susceptibility of schizophrenia.
Theoretical bases of how different SNPs affect pathophysiology of psychiatric disorders are not well established. However, considering preexisting results will help understand our results. 196G/A is known to modulate expression of BDNF protein. Structurally, 196G/A allele difference results in different brain volumes and A allele in smaller prefrontal, dorsolateral volumes compared to G allele [25]. 11757G/C is also associated with structural differences, frontal gray matter volume variation, and cerebellar hemisphere white and gray matter volumes in imaging studies. Considering these results, susceptibility of schizophrenia depending on SNP difference can be explained partially. 11757G/C allele variance, for example, results in difference in frontal volume, especially in smaller frontal volume, and this hypofrontality also contributes to schizophrenia. A few evidence for impact of different SNPs in brain volume or BDNF level exist, however, it was not a distinct feature in etiology of schizophrenia.
SNP genotypes could not predict clinical severity in schizophrenia patients. In our study, the 2 SNP genotypes did not show significant differences in symptom severity scales. This result is in agreement with a previous study showing that 196G/A genotype is related with the presence of symptoms rather than severity of symptoms [6]. Affective symptoms, anxiety and depressive symptoms showed an association with carrying the A allele [26]. However, a previous study showed the G/A genotype for 196G/A was related to earlier onset of disease and the G/G genotype was associated with severe symptoms [27]. Future studies are needed to clarify such inconsistent results.
In the present study, the existence of A allele in 196G/A correlated with a personal history of suicide attempts. Both postmortem study [28] and study with living patients [29] showed the presence of the A allele was associated with susceptibility to suicide in depressed patients. In the present study, the A allele also predicted suicide attempts in schizophrenia patients regardless of different diagnoses.
Considering function of SNP 196G/A in BDNF expression may help understand our results. As mentioned above, 196G/A expression plays a role in modulating production and secretion of BDNF protein in brain. Especially, A allele in 196G/A is related to lowering production of BDNF in brain compared to G allele [30]. Low BDNF level causes disruption in the neuronal survival and plasticity of serotonergic and dopaminergic neurons, and, such disruption can result in suicidality [31]. Low brain BDNF level in hippocampus and prefrontal cortex is also shown to be related with suicidal behavior [29]. Recently, suicide-attempted patients had lower levels of BDNF, even in serum [32]. Furthermore, A allele in 196G/A was also dominant among patients with anxiety-related personality traits, and it brings higher frequency of suicidal behaviors [29]. All these impacts of BDNF protein expression on neurobiological pathways and personality traits would explain specific BDNF SNP and its relation to suicide behaviors.
Previous studies also confirm our results. A allele in 196G/A and its relation with the suicide behavior has been well studied. A/A seemed to be the most related with history of suicide compared to G/A or G/G [33]. Like our study, A carrier showed relation with suicide attempt in schizophrenia patients. Among A carriers, previous history of childhood trauma or stressful life events even raised the risk for suicide attempt [28]. Psychiatric-related dysfunction in the patients also raised risk for suicide attempt among A carriers [34].
Suicide attempt rate ranges from 18% to 55% among schizophrenia patients [35]. Similar to many other risk factors, such as younger age [36], male gender [37], earlier age at onset [38], being unmarried [39], presence of the A allele can be used as risk factors for suicide in schizophrenia patients. Verifying susceptibility to suicide in a clinical setting will help manage these patients which could be life-saving.
Overall results also suggest that BDNF gene polymorphism cannot directly predict susceptibility of disease, but rather predict the specific symptom entity among psychiatric disorders. A allele in 196G/A and its relation with affective symptoms in schizophrenia [26] for instance. In our study, we also found that the BDNF gene polymorphism could not predict susceptibility to the disease itself, but rather predict relation with specific clinical features among schizophrenia patients. Similar results were suggested in study of the depression [40]. It was also found that specific alleles and genotypes were related with severe depressive symptoms, rather than diagnosis of depression. These results can partly explain that the BDNF polymorphism is specifically related with the neuropsychiatric symptoms. Further study to support current result is required.
This study had several limitations. The number of patients and controls was relatively small. Also, our results do not include quantitative information about the BDNF level in both each brain sites and whole body serum, but only genotypes and gene alleles. Few of our analysis didn’t have done corrections for multiple comparisons. Lastly, our study included only Korean patients and limited ethnic diversity could have produced biased results. Regardless of these limitations, this study is valuable for examining clinical variables and their relationship with BDNF SNP genotype. Future studies overcoming our study limitations will expand the understanding of the neurobiology and etiology of schizophrenia.
Acknowledgements
This paper was supported by Korea University.
