 |
 |
- Search
| Psychiatry Investig > Volume 19(3); 2022 > Article |
|
Abstract
Objective
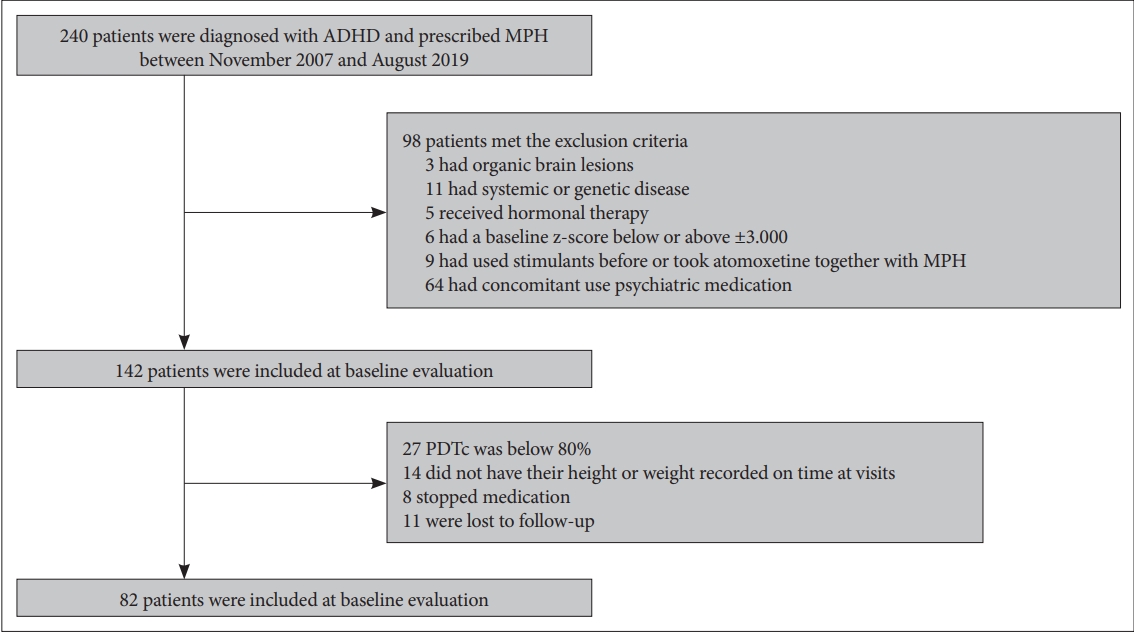
Methods
Results
Notes
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Wan Seok Seo. Data curation: Yoojeong Lee, Nayeong Kong, San Koo. Formal analysis: Dai Seg Bai, Hyeonseok Jeong. Funding acquisition: Wan Seok Seo, Hee jin Kim. Investigation: Hee jin Kim. Methodology: Wan Seok Seo, Nayeong Kong. Project administration: Wan Seok Seo. Resources: Hee jin Kim. Software: Wan Seok Seo, Yoojeong Lee. Supervision: Wan Seok Seo. Validation: Wan Seok Seo, Yoojeong Lee. Visualization: Wan Seok Seo, Yoojeong Lee. WritingŌĆöoriginal draft: Yoojeong Lee, Nayeong Kong, San Koo. WritingŌĆöreview & editing: Wan Seok Seo.
Funding Statement
This study was supported by a Yeungnam University Research Grant in 2018.
Figure┬Ā1.
Figure┬Ā2.

Table┬Ā1.
Table┬Ā2.
| Baseline | 6 months | 12 months | 18 months | 24 months | p* | |
|---|---|---|---|---|---|---|
| Height z-score | 0.002┬▒1.059 | -0.100┬▒1.082 | -0.159┬▒1.059 | -0.158┬▒1.056 | -0.186┬▒1.068 | 0.001 |
| ŌĆāpŌĆĀ | <0.001 | <0.001 | <0.001 | 0.001 | ||
| Weight z-score | 0.104┬▒0.945 | -0.155┬▒0.877 | -0.256┬▒0.969 | -0.278┬▒0.935 | -0.301┬▒0.986 | <0.001 |
| ŌĆāpŌĆĀ | <0.001 | <0.001 | <0.001 | <0.001 | ||
| BMI z-score | 0.138┬▒0.936 | -0.150┬▒0.872 | -0.250┬▒0.960 | -0.245┬▒0.976 | -0.293┬▒0.972 | 0.001 |
| ŌĆāpŌĆĀ | <0.001 | <0.001 | <0.001 | <0.001 |
Table┬Ā3.
| Measures by sex | Baseline | 6 months | 12 months | 18 months | 24 months | p* |
|---|---|---|---|---|---|---|
| Height z-score | 0.095 | |||||
| ŌĆāMale (N=64) | 0.097┬▒0.968 | 0.008┬▒1.001 | -0.512┬▒0.994 | -0.049┬▒0.990 | -0.930┬▒1.019 | |
| ŌĆāFemale (N=18) | -0.338┬▒1.311 | -0.486┬▒1.289 | -0.544┬▒1.219 | -0.545┬▒1.217 | -0.515┬▒1.201 | |
| Weight z-score | 0.017 | |||||
| ŌĆāMale (N=64) | 0.231┬▒0.928 | -0.011┬▒0.828 | -0.120┬▒0.889 | -0.129┬▒0.853 | -0.211┬▒0.902 | |
| ŌĆāFemale (N=18) | -0.348┬▒0.887 | -0.667┬▒0.875 | -0.741┬▒1.110 | -0.671┬▒1.120 | -0.623┬▒1.213 | |
| BMI z-score | 0.044 | |||||
| ŌĆāMale (N=64) | 0.247┬▒0.982 | -0.028┬▒0.884 | -0.137┬▒0.935 | -0.157┬▒0.946 | -0.229┬▒0.946 | |
| ŌĆāFemale (N=18) | -0.248┬▒0.628 | -0.586┬▒0.682 | -0.651┬▒0.966 | -0.562┬▒1.042 | -0.519┬▒1.054 |
Table┬Ā4.
| Measures by baseline age | Baseline | 6 months | 12 months | 18 months | 24 months | p* |
|---|---|---|---|---|---|---|
| Height z-score | 0.798 | |||||
| ŌĆā<9 y (N=57) | -0.055┬▒1.051 | -0.157┬▒1.104 | -0.196┬▒1.080 | -0.199┬▒1.064 | -0.247┬▒1.073 | |
| ŌĆā9-12 y (N=19) | 0.140┬▒1.184 | 0.041┬▒1.111 | -0.043┬▒1.071 | -0.040┬▒1.110 | -0.022┬▒1.073 | |
| ŌĆāŌēź12 y (N=6) | 0.102┬▒0.805 | -0.016┬▒0.869 | -0.180┬▒0.956 | -0.140┬▒0.938 | -0.122┬▒1.138 | |
| Weight z-score | 0.928 | |||||
| ŌĆā<9 y (N=57) | 0.117┬▒0.932 | -0.115┬▒0.875 | -0.278┬▒0.944 | -0.279┬▒0.849 | -0.421┬▒0.863 | |
| ŌĆā9-12 y (N=19) | 0.107┬▒1.053 | -0.213┬▒0.962 | -0.243┬▒1.011 | -0.197┬▒1.075 | -0.102┬▒1.060 | |
| ŌĆāŌēź12 y (N=6) | -0.034┬▒0.846 | -0.352┬▒0.696 | -0.089┬▒1.235 | -0.110┬▒1.379 | 0.206┬▒1.655 | |
| BMI z-score | 0.990 | |||||
| ŌĆā<9 y (N=57) | 0.184┬▒0.946 | -0.068┬▒0.871 | -0.260┬▒0.949 | -0.267┬▒0.925 | -0.413┬▒0.853 | |
| ŌĆā9-12 y (N=19) | 0.090┬▒0.923 | -0.290┬▒0.860 | -0.275┬▒0.864 | -0.216┬▒0.928 | -0.110┬▒0.908 | |
| ŌĆāŌēź12 y (N=6) | -0.144┬▒0.979 | -0.486┬▒0.933 | -0.078┬▒1.453 | -0.135┬▒1.652 | 0.278┬▒1.871 |
REFERENCES







