 |
 |
- Search
| Psychiatry Investig > Volume 20(4); 2023 > Article |
|
Abstract
Objective
Methods
Results
Conclusion
Notes
Availability of Data and Material
Data is available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Author Contributions
Data curation: Qifang Lu, Yanming Wang, Li Huang, Xiaoxiao Wang. Formal analysis: Qifang Lu, Jin Chen, Zhoufan Jiang. Project administration: Qifang Lu, Xiaoxiao Wang. Visualization: Qifang Lu, Jin Chen, Zhoufan Jiang. Writing—original draft: Qifang Lu, Xiaoxiao Wang. Writing—review & editing: Jin Chen, Benedictor Alexander Nguchu, Shishuo Chen, Bensheng Qiu, Xiaoxiao Wang.
Funding Statement
This work was supported by the National Natural Science Foundation of China (grant nos. 81701665, 21876041), the Fundamental Research Funds for the Central Universities (WK5290000002), the University Synergy Innovation Program of Anhui Province (GXXT-2021-003), the scientific research project of Health Commission of Anhui Province (AHWJ2021a034), the provincial education research project of Department of Education Anhui Province (2020jyxm0945), the innovation team project of Anhui Medical College (YZ2020TD005).
Figure 1.
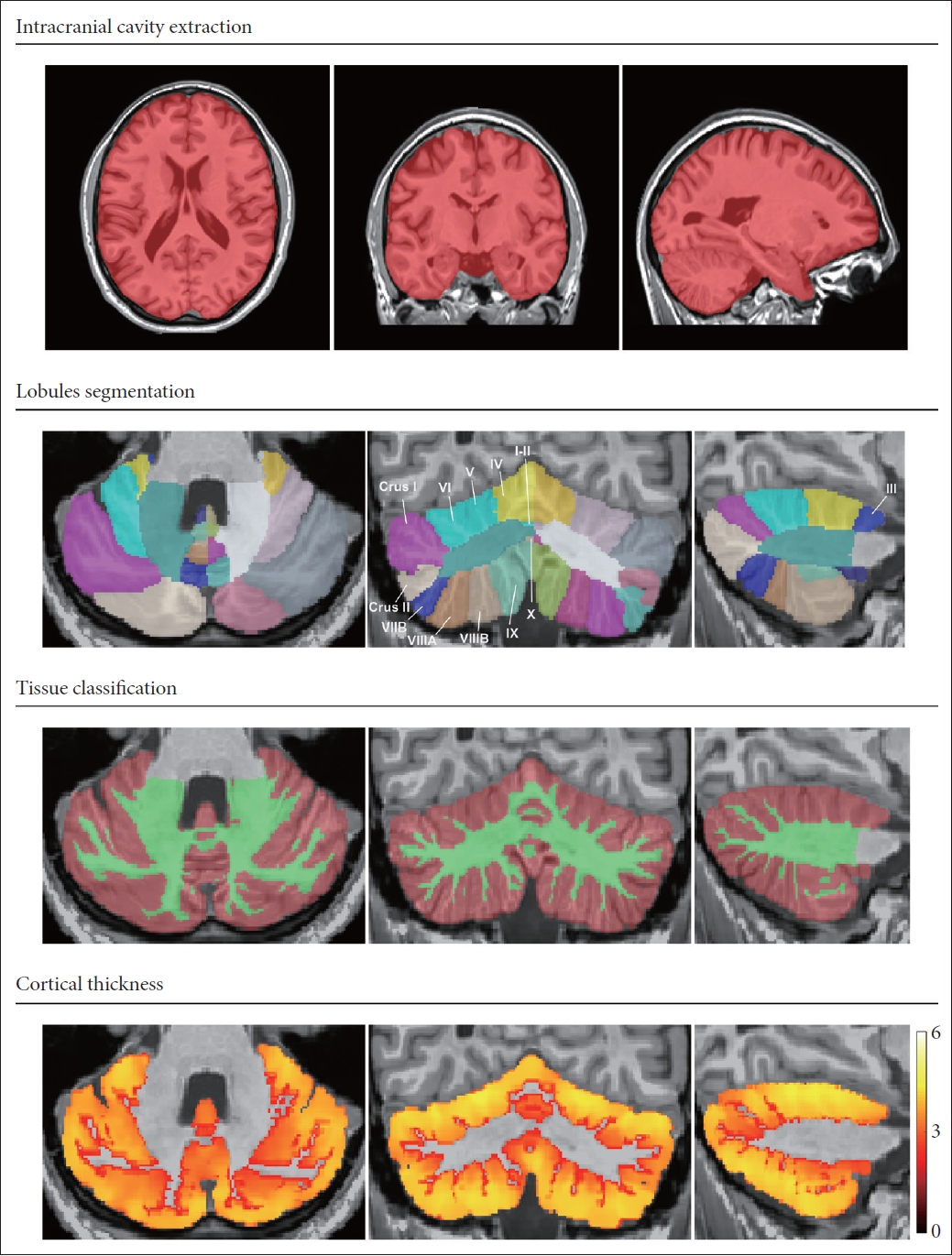
Figure 2.

Figure 3.

Table 1.
| ASD (N=75) | TD (N=97) | p | |
|---|---|---|---|
| Age (yr) | 11.86±2.78 | 12.53±2.99 | 0.129† |
| Gender (male:female) | 1900-01-02 16:11 | 1900-01-03 0:25 | 0.076‡ |
| FIQ* | 107.02±17.13 | 111.94±13.76 | 0.065† |
| ADI-R_ Social total | 19.94±5.72 | N/A | N/A |
| ADI-R_Verbal total | 16.2±4.60 | N/A | N/A |
| ADI-R_RRB total | 5.96±2.75 | N/A | N/A |
| ADI-R_Onset total | 3.35±1.47 | N/A | N/A |
Values are presented as mean±standard deviation. The ADI-R score statistics are for the 54 individuals in the ASD group for whom scores were recorded. ADI-R_ Social total: reciprocal social interaction subscore total for ADI-R; ADI-R_Verbal total: abnormalities in communication subscore total for ADI-R; ADI-R_RRB total: restricted, repetitive, and stereotyped patterns of behavior subscore total for ADI-R; ADI-R_Onset total: abnormality of development evident at or before 36 months subscore total for ADIR.
Table 2.
| Normalized ortical thickness | Group | Mean | SD | SEM | p |
|---|---|---|---|---|---|
| VI right | ASD | 4.145 | 0.228 | 0.026 | 0.006* |
| TD | 4.234 | 0.194 | 0.020 | ||
| VI left | ASD | 4.257 | 0.208 | 0.024 | 0.017* |
| TD | 4.328 | 0.178 | 0.018 | ||
| Crus I right | ASD | 4.029 | 0.226 | 0.026 | 0.016* |
| TD | 4.106 | 0.192 | 0.020 | ||
| Crus I left | ASD | 4.018 | 0.227 | 0.026 | 0.008* |
| TD | 4.111 | 0.222 | 0.023 | ||
| X left | ASD | 2.063 | 0.338 | 0.039 | 0.01* |
| TD | 2.201 | 0.349 | 0.035 |
REFERENCES







