 |
 |
- Search
| Psychiatry Investig > Volume 21(1); 2024 > Article |
|
Abstract
Objective
Methods
Results
Conclusion
Supplementary Materials
Supplementary Table 1.
Supplementary Table 2.
Notes
Availability of Data and Material
The datasets generated or analyzed during the current study are not publicly available due to Patient Data Management Protocol of Yeouido St. Mary’s Hospital but are available from the corresponding author on reasonable request.
Conflicts of Interest
Yoo Hyun Um and Hyun Kook Lim, a contributing editor of the Psychiatry Investigation, were not involved in the editorial evaluation or decision to publish this article. All remaining authors have declared no conflicts of interest.
Author Contributions
Conceptualization: Sunghwan Kim. Data curation: Sunghwan Kim, Hyeonsik Yang, Hyunji Lee. Formal analysis: Sunghwan Kim, Hyeonsik Yang, Hyunji Lee. Funding acquisition: Hyun Kook Lim. Investigation: Sunghwan Kim, Sheng-Min Wang, Dong Woo Kang, Yoo Hyun Um. Methodology: Sunghwan Kim, Hyeonsik Yang, Hyunji Lee. Project administration: Regina EY Kim, Donghyeon Kim, Hyun Kook Lim. Resources: Donghyeon Kim, Hyun Kook Lim. Software: Sunghwan Kim, Hyeonsik Yang, Hyunji Lee, Regina EY Kim. Supervision: Chang Uk Lee, Hyun Kook Lim. Validation: Sheng-Min Wang, Dong Woo Kang, Yoo Hyun Um. Visualization: Sunghwan Kim, Hyeonsik Yang, Hyunji Lee. Writing—original draft: Sunghwan Kim, Hyeonsik Yang, Hyunji Lee. Writing—review & editing: Sheng-Min Wang, Hyun Kook Lim.
Funding Statement
This research was supported by the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI) and Korea Dementia Research Center (KDRC), funded by the Ministry of Health & Welfare and Ministry of Science and ICT, Republic of Korea (grant number: HU20C0315).
Figure 1.
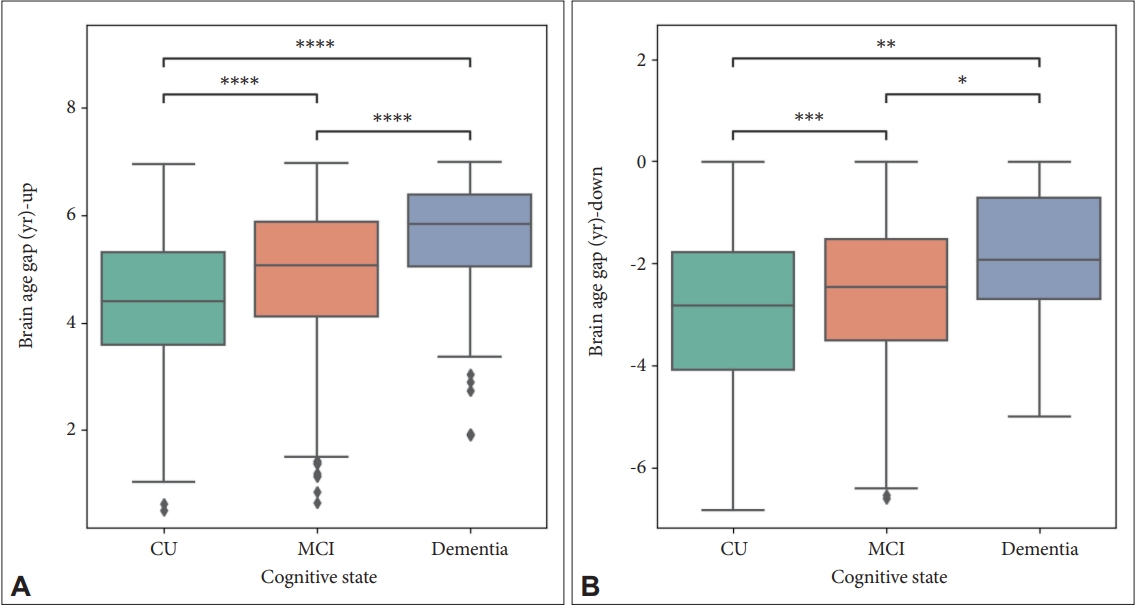
Figure 2.
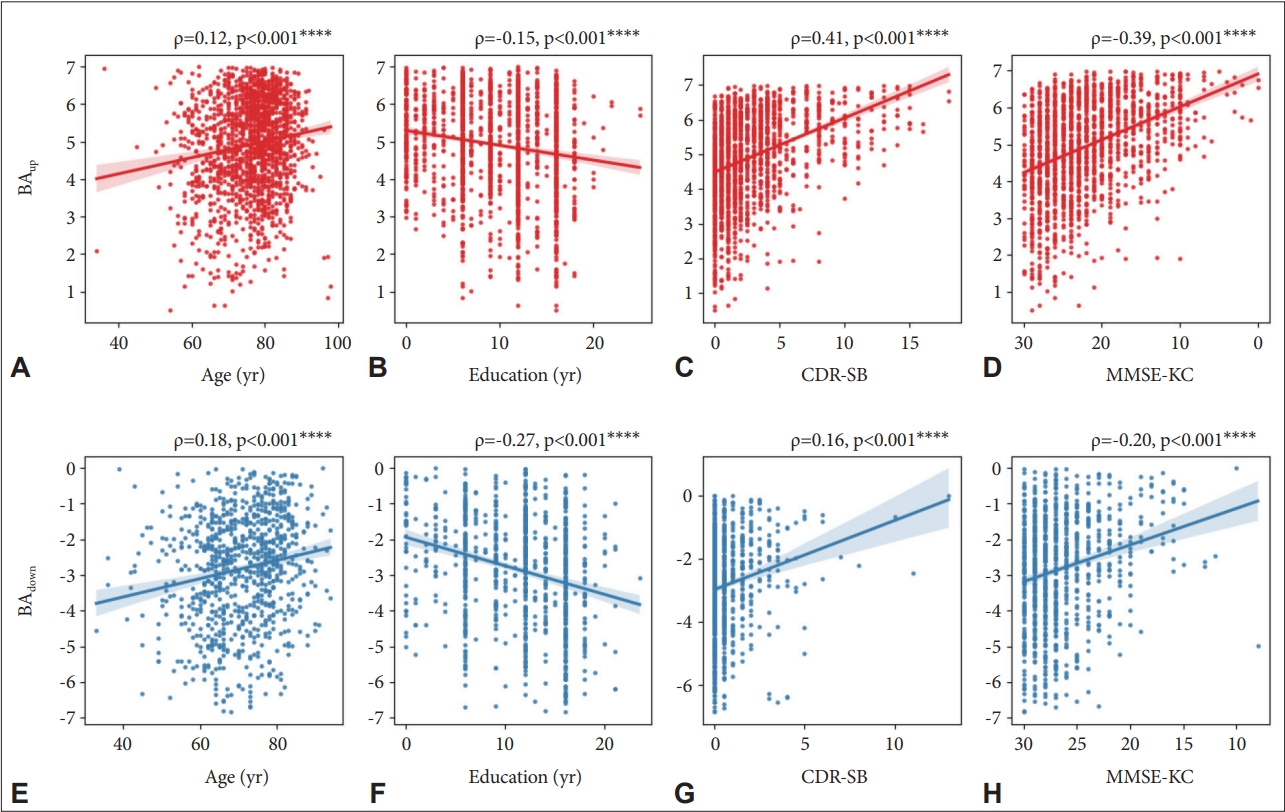
Table 1.
| Characteristics | CU | MCI | Dementia | Total | Statistics | p |
|---|---|---|---|---|---|---|
| Sex | χ2=2.62 | 0.27 | ||||
| Female | 690 (27.0) | 829 (32.4) | 274 (10.7) | 1,793 (70.0) | ||
| Male | 319 (12.5) | 344 (13.4) | 104 (4.1) | 767 (30.0) | ||
| ApoE4 | χ2=57.42 | 0.001* | ||||
| Carrier | 233 (9.1) | 376 (14.7) | 164 (6.4) | 773 (30.2) | ||
| Noncarrier | 776 (30.3) | 797 (31.1) | 214 (8.4) | 1,787 (69.8) | ||
| Age (yr) | 70.58±9.35 | 75.56±8.11 | 79.19±8.03 | 74.13±9.15 | F=167.80 | <0.001* |
| Education (yr) | 11.04±4.81 | 9.62±5.35 | 9.02±5.51 | 10.10±5.23 | F=29.97 | <0.001* |
| MMSE-KC | 27.32±2.34 | 23.42±3.86 | 15.61±5.13 | 23.80±5.25 | F=1,496.31 | <0.001* |
| CDR | 0.11±0.21 | 0.47±0.15 | 1.21±0.51 | 0.44±0.44 | F=2,495.19 | <0.001* |
| CDR-SB | 0.27±0.50 | 1.84±1.26 | 7.22±3.12 | 2.02±2.73 | F=2,942.21 | <0.001* |
The data are presented as mean±SD format for continuous variables and “counts (proportion in percentage)” format for categorical variables.
CU, cognitively unimpaired; MCI, mild cognitive impairment; MMSE-KC, Mini-Mental State Examination in the Korean version of Consortium to Establish a Registry of Alzheimer’s Disease Assessment Packet; CDR, clinical dementia rating; CDR-SB, clinical dementia rating sum-of-boxed; SD, standard deviation
Table 2.
| Number of ROIs (<5%) | CU (yr) | MCI (yr) | Dementia (yr) |
|---|---|---|---|
| 0 | -2.13±2.62 | -1.04±3.05 | 0.96±3.36 |
| 1 | 3.36±2.49 | 4.19±1.14 | 4.63±0.96 |
| 2 | 3.66±2.57 | 4.53±1.44 | 5.17±0.80 |
| 3 | 4.98±0.90 | 5.38±0.72 | 5.47±0.83 |
| 4 | 5.44±0.90 | 5.54±0.78 | 5.66±0.68 |
| 5 | 5.72±0.66 | 5.87±0.65 | 6.08±0.64 |
| 6 | 5.88±0.65 | 5.94±0.64 | 6.10±0.62 |
| 7 | 5.76±0.57 | 6.39±0.50 | 6.22±0.50 |
| 8 | 6.52* | 6.10±0.55 | 6.42±0.51 |
| 9 | - | - | - |
| 10 | 5.52* | 6.58* | 6.70±0.42 |
| 11 | - | - | - |
| 12 | - | - | - |
| All | 0.10±3.89 | 2.95±3.54 | 5.26±1.91 |
REFERENCES







