INTRODUCTION
The occurrence of depression increases dramatically during adolescence.1 Furthermore, all over the world, more than 1/4 of adolescents are obese which is usually measured by body mass index (BMI).2 Some researchers have proposed that the BMI may influence depressive symptoms through body image.3 In empirical research, some studies found that obese and overweight adolescents were more likely to become depressed than normal ones,45 whereas Wardle et al.6 found that low weight was related to more depressive symptoms. Recently, some researchers have proposed that a u-shaped relationship may explain these mixed findings.78 Additionally, in previous research, Kress et al.'s9 study indicated that no differences existed between overweight and normal weight adolescents in their depressive symptoms.
Moreover, mono-amine-oxidase type A (MAOA) gene has been suggested to be a candidate gene that is associated with depression.10 The MAOA gene has a functional polymorphism (2-repeat, 3-repeat, 3.5-repeat, 4-repeat, and 5-repeat),11 which is based on a 30-bp insertion or deletion. Until now, the empirical findings about the relationship between the MAOA gene polymorphism and depressive symptoms have been mixed. Some studies indicated that the 4-repeat allele carriers were more likely to become depressed than the 3-repeat allele carriers,1213 while some other studies did not find a significant relationship between the MAOA gene polymorphism and depressive symptoms.1415
However, to our knowledge, no study has tested whether the Western findings about the effects of the BMI and the MAOA gene polymorphism can be generalized to Asia adolescents. Therefore, the present study seeks to examine the joint effects of the BMI and the MAOA gene polymorphism on depressive symptoms by using a Chinese sample. It is expected that 1) low weight and obese adolescents both show more depressive symptoms; 2) the 4-repeat allele carriers have more depressive symptoms; 3) the MAOA gene polymorphism moderates the relationship between the BMI and depressive symptoms. In previous research, several studies found that gender contributed to depressive symptoms.161718 Hence, we control the effects of gender on depressive symptoms, when analyzing the joint effects of the BMI and the MAOA gene polymorphism on depressive symptoms.
METHODS
Participants and procedures
In this study, two independent samples were used. One sample (sample 1) consisted of 211 grade 10 students (107 girls). Their mean age was 16.48 years (SD=0.56). The other sample (sample 2) was used to replicate the findings of sample 1. This sample included 235 grade 10 (102 females) from another three high schools. The mean age of these students was 16.59 years (SD=0.67). In both samples, students' depressive symptoms were measured and their DNA was collected during classroom time. Their weight and height at grade 10 were measured by trained nurses at school. We received ethical approval from our institution and informed consent was gotten from each participant and one of his/her parents.
Measures
Depressive symptoms
Adolescents' depressive symptoms were assessed by using a 10-item shortened version of the Children's Depressive Inventory.19 A three-point scale was used, with anchors 1: Never and 2: Always. In this study, the internal consistencies of this scale were acceptable at both samples (╬▒=0.78 at sample 1 and ╬▒=0.76 at sample 2).
DNA collection and genotyping
DNA was extracted from students' cheek cells. Polymerase chain reaction (PCR) was used to amplify the DNA fragment. The primer sequences were 5'-GAACGGACGCTCCATTC GGA-3' (forward) and 5'-ACAGCCTGACCGTGGAGA AG-3' (reverse). The following cycling conditions were used: initial denaturation at 95Ōäā for 5 minutes, followed by 35 cycles at 94Ōäā for 30 seconds, 58Ōäā for 30 seconds, and 72Ōäā for 40 seconds, and finally elongation at 72Ōäā for 10 minutes. The length of PCR products was separated by using a 2% agarose gel electrophoresis.20
RESULTS
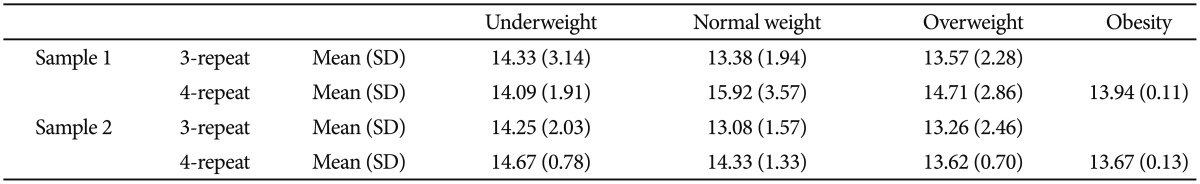
In the sample 1, the results indicated that 107 students were the 3-repeat genotype (3 or 3/3), 54 students were the 4-repeat genotype (4 or 4/4), and 48 students were heterogeneous genotype (3/4). In the sample 2, there were 124 students with only the 3-repeat allele, 68 students with only the 4-repeat allele, and 40 students with the 3/4-repeat genotype. The 2-repeat (1 participant in each sample) and 5-repeat (1 participants in the sample 1 and 2 participants in the sample 2) allele carriers were found as well. As the MAOA gene is on the X chromosome, the phenomenon of gene suppression for females can happen. Similar to the prior studies,20 the students with the heterogeneous genotype were eliminated in the next analysis. Moreover, in the sample 1, 26 students were underweight (BMI <18.5 kg/m2), 151 were normal weight (BMI=18.5-24.9 kg/m2), 31 were overweight (BMI=25.0-29.9 kg/m2), and 3 were obesity (BMI >30.0 kg/m2).8 In the sample 2, 37 students were underweight, 174 were normal weight, 24 were overweight, and 3 were obesity.
Means and standard deviations of students' depressive symptoms in both samples are shown in Table 1. ANOVAs revealed that the main effects of the MAOA gene polymorphism on depressive symptoms were significant [for sample 1, F (1, 153)=4.11, p<0.05, ╬Ę2=0.03; for sample 2, F (1, 184)=3.97, p<0.05, ╬Ę2=0.02]. However, the main effects of the BMI [for sample 1, F (3, 153)=0.59, ns; for sample 2, F (3, 184)=2.14, ns] and the interaction of the MAOA gene polymorphism and the BMI [for sample 1, F (2, 153)=2.24, ns; for sample 2, F (2, 184)=1.28, ns] on depressive symptoms were not significant. During analysis, gender was treated as a control variable.
DISCUSSION
The present study significantly contributes to theory and practices by examining the joint effects of the BMI and the MAOA gene polymorphism on depressive symptoms. Furthermore, two independent samples is another strength of the present study.
In this study, our data partly supported our hypothesis. First, by using a Chinese sample, our data indicated that the MAOA gene as a genetic factor directly contributed to depressive symptoms.13 In line with some previous Western studies,13 the present study confirms that adolescents with the 4-repeat allele show more depressive symptoms than the 3-repeat allele carriers. Our findings suggest that the links between the MAOA gene polymorphism and depressive symptoms can be generalized to Chinese adolescents. Second, our data showed that Chinese adolescents' BMI was not significantly related to their depressive symptoms. The present findings provide supportive evidence to the previous findings that showed no differences in depressive symptoms between overweight and normal weight adolescents.9 The present findings imply that the Western findings about the significant effects of the BMI on depressive symptoms7821 can not be generalized to Chinese adolescents. Hence, to change BMI was not a effective way to reduce Chinese adolescents' depressive symptoms. Third, our data did not show significant interaction of the MAOA gene polymorphism and the BMI. These findings suggest that the MAOA gene polymorphism does not moderate the relationship between the BMI and depressive symptoms. Taken together, our findings suggest that the MAOA gene polymorphism only directly contributes to adolescents' depressive symptoms.
In this study, there are some limitations that could be improved in further research. For instance, the present study was a cross-sectional one. Further research may use a longitudinal design to replicate the present findings. Furthermore, four types of alleles were found in our Chinese sample. However, the number of participants with the 2-repeat and 5-repeat allele was very small. Further research may use a larger sample to examine the joint effects of such alleles and the BMI on depressive symptoms.