|
 |
- Search
| Psychiatry Investig > Volume 7(1); 2010 > Article |
Abstract
Objective
The core deficit of attention deficit hyperactivity disorder (ADHD) is associated with frontal cortex and related circuitry. Children with ADHD and a medication history have shown atypical brain activation in prefrontal and striatal brain regions during cognitive challenge. We investigated two cognitive control operations such as interference suppression (IS) and response inhibition (RI) in children with ADHD. We also assessed the brain functions affected by the methylphenidate (MPH) effect by comparing the blood-oxygen level dependent (BOLD) signals in ADHD children on and off medication.
Methods
Eight children (9-11 years of age) with combined-type ADHD underwent rapid event-related functional magnetic resonance imaging (fMRI) during performance of a modified flanker task. Two fMRI (3.0 T) scans were conducted with a one week interval-one with MPH treatment and the other without. Functional maps were generated through group averaging and performance-based correlational analyses.
Results
Performances of the two cognitive control operations did not differ significantly between on-MPH and off-MPH status other than the reaction time to incongruent stimuli in ADHD children. In those affected by MPH treatment, an increased activation in the right prefrontal cortex during incongruent task was observed relative to a neutral trial in children with ADHD.
Conclusion
On the treatment of MPH, the ADHD children exhibited increased activation of the right frontal cortex during interference suppression. This finding suggested that MPH affected the right frontal cortex in ADHD compensating for a reduced level of interference suppression. Future studies will be required to ascertain the MPH effect of cognitive brain regions among large number of children with ADHD.
Attention deficit hyperactivity disorder (ADHD) is the most common developmental disorder of childhood,1 and its various cognitive and behavioral manifestations can be explained by a deficit in the frontal cortex and related circuitry.2 Several cognitive models have proposed that reduced response inhibition (RI) is a cardinal symptom of ADHD.1 Cognitive control, the ability to voluntary constrain actions in a goal-directed manner, comprises at least two operations, RI and interference suppression (IS). Measured by error of commission on cue-guided tasks in the inhibition of a prepotent response, RI is reduced in subjects with ADHD.3,4 Similarly, IS, which can be measured by erroneous or slower responses to target stimulus due to of interference from competing responses, is also reduced in subjects with ADHD.5 However, neural substrates of reduced cognitive control in ADHD are not yet fully understood.
Generally, when children grow into adolescences, RI and IS improve6,7 in accordance with myelination and synaptic organization,8,9 and in accordance with functional activation of the frontal cortex.10,11 However, children with ADHD show delayed or abnormal maturation of the frontal cortex and related circuitry leading to inefficient cognitive control behaviors.4 Methylphenidate (MPH), used as the first line therapy for ADHD patients, is known to significantly improve cognitive control function with respect to RI; however, with only minimal effect on IS.5
Structural MRI studies have demonstrated abnormally reduced right frontal lobes,11 as well as similarly affected striatal12 and cerebellar13 structures in ADHD subjects. Causal models of ADHD have long implicated dysfunction in the frontostriatal networks supporting executive function, a hypothesis that can be examined systematically using functional neuroimaging.12,14 Previous functional imaging studies in the course of the go/no-go have shown abnormalities in frontal lobe activation in children and adolescents with ADHD.4 Additionally, children with ADHD together with their medication history have shown atypical brain activation in prefrontal and striatal brain regions during cognitive challenge.15 To the best of our knowledge, few studies have been done in children with ADHD in Korea using fMRI. In this study, we investigated the neural basis of two cognitive control operations, IS and RI, among children with ADHD. We also assessed the brain regions affected by the MPH effect by comparing the blood-oxygen level dependent (BOLD) signals in ADHD children either on or off medication.
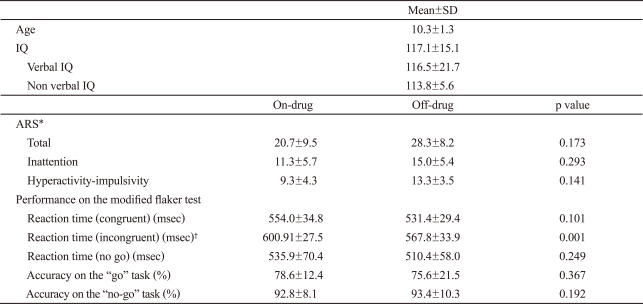
This study was conducted in eight right handed ADHD boys (age: 10.3┬▒1.3, elementary school 4-6th grade) who exhibited a good treatment response to MPH. All patients were put into the ADHD category according to DSM-IV16 and the ADHD Rating Scale-IV (ARS)17 after interviews with child and adolescent psychiatrist. Diagnoses were confirmed by another psychiatrist according to Kiddie Schedules for Affective Disorder and Schizophrenia-Present and Lifetime version (KSADS-PL).18 Exclusion criteria were those under an IQ of 85 as per the Wechsler Intelligence Scale for Children (K-WISC-III) test,19 and those who had history or evidence of neurological disorders or Axis 1 psychiatric disorders. Study objectives and methods were fully explained to recruited patients and parents and verbal assent and written consent were obtained.
If needed, prior to MRI scan, an explanation of the procedures and equipment used in fMRI was provided. Fees for two fMRI scanning were paid by the investigators. This study protocol was approved by the Institutional Review Board (IRB) of Chung Ang University Hospital, South Korea.
The first MRI scan was performed while the patients were medicated, and the second MRI scan was done after the medication had been intentionally discontinued for 1 week as previously suggested by Shafritz et al.20 and Zang et al.21 Every attempt was made to maintain general clinical treatment settings. Patients were instructed to take their medication at 8 a.m. and the fMRI was performed at 2 p.m. when the maximum concentration had been attained. Five ADHD patients were taking Concerta® (27-45 mg) and other three others were taking Metadate CD® (30-40 mg). Both fMRI scans were performed at the same time of day. The subjects viewed the projected, computer-controlled paradigm on a screen at the head of the scanner via a 45° angled mirror, affixed to the MRI head coil. They used their right hand to hold a button and responded by pressing it with their index and middle fingers. To minimize head movement, memory form fit to each subject was used.
Subjects performed two runs of a modified Eriksen flanker task (Figure 1)6 using E-prime (Psychology Software Tools, Pittsburgh, PA, USA). Each trial consisted of a display comprised of a central arrow with two flanking stimuli on either side. Subjects were instructed to press one of two buttons indicating the direction of the central arrow (left/right) while ignoring the flanking stimuli. On congruent and incongruent trials, the flankers were arrows pointing in the same or different directions, respectively in relation to the central arrow. On neutral trials, the flankers were diamonds that were not associated with a response. On no-go trials, the flanking stimuli signals were Xs, indicated that subjects should have withheld their response. Trials were presented in a rapid event-related design10 with a 3-second intertrial interval. In each trial, the display (800 msec) was followed by a blank screen (300 msec) and then a fixed stimulus (1600 msec); intertrial interval was 300 msec. Subjects practiced about 40 times for about 2 minutes prior to conducting the actual test, which consisted of 252 trials all of which took 12 minutes and 36 seconds.
Prior to the MRI scans, a subject's head was fixed and confirmed as to whether the subject could see the screen. A 3-T MRI (Philips Achieva 3.0T X-series) was used to acquire T1-weighted flow compensated spin-echo anatomical images (TR=500 msec; minimum TE) in 16 contiguous 7-mm axial slices, parallel to the plane of the anterior commissure-posterior commissure. Functional acquisition included the same slices with a T2*-sensitive gradient echo spiral pulse sequence with parameters of TR=1,000 msec, TE=30 msec, a field of view=23 cm, a flip angle=90┬░ and an 80├Ś80 matrix.
Using SPM2 (Wellcome Department of Cognitive Neurology, London, UK), images were corrected for differences in slice acquisition time and motion. All subjects displayed less than 1 mm of motion in x, y, and z directions throughout the course of each scan. Images were normalized into a standard space22 and interpolated to 2├Ś2├Ś2 mm cubic voxels. Normalized image volumes were spatially smoothed (8-mm full width at half-maximum Gaussian kernel) and temporally filtered (low-pass filter: 4-msec Gaussian; high-pass filter: calculated on the basis of trial frequency). fMRI responses were modeled using a canonical hemodynamic response function. For each subject, activation maps were generated using linear contrasts identifying regions that were more active during incongruent relative to neutral trials and during no-go relative to neutral trials. All analyses were generated by using a voxel-level height threshold of p<0.05 uncorrected for multiple comparisons as well as a spatial extent threshold of 30 voxels.
Demographic and clinical characteristics of subjects are shown in Table 1. The ARS score of subjects at baseline was 36.2±8.3 (inattention 19.8±3.2, hyperactivity-impulsivity 16.3±5.9) and Clinical Global Scale-Severity (CGI-S) was over 4 (4.5±0.8). After three months of treatment, those with CGI-S and CGI-improvement (CGI-I) scores less than 2 were defined as good responders and were included in this study. The ARS score for on-MPH was 20.7±9.5 (inattention 11.3±5.7, hyperactivity-impulsivity 9.3±4.3) and that for off-MPH was 28.3±8.2 (inattention 15.0±5.4, hyperactivity-impulsivity 13.3±3.5). There were no significant differences in cognitive data during the modified Eriksen flanker task between on and off MPH, including reaction time of congruent stimuli and accuracy rate on go/no-go tasks. But there was a significant difference in the reaction time of incongruent stimuli between on and off treatment (p=0.01). Average MPH doses during first scan was 34.2±7.5 mg/day for Concerta® and 36.7±5.8 mg/day for Metadate CD®.
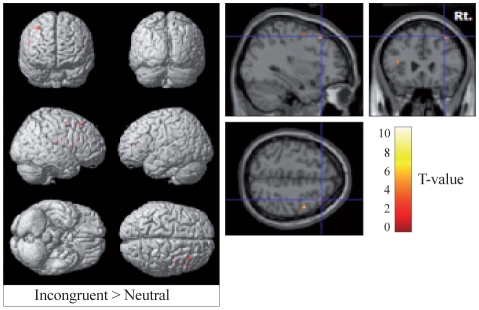
As seen Table 2 and Figure 2, ADHD children exhibited activation of the right frontal lobe, and in particular, the superior frontal gyrus (Brodmann's area 8), precentral gyrus, middle frontal gyrus (Brodmann's area 46), and inferior frontal gyrus, at a significance threshold of uncorrected p<0.001 or 0.005 during incongruent relative to neutral trial while undergoing MPH treatment. There were no significant differences in neural correlates of RI between on and off MPH.
This study was an fMRI pilot study conducted in 4-6th grade elementary school children using an event-related design. The authors believe this study to be the first fMRI study conducted in Korean children with ADHD using such a design. From the study results, the suppression of the interfering response was associated with an increased activation of the right frontal lobe in medicated ADHD children. This finding suggested that MPH affected the activation of the right frontal cortex, which correlates with improvement of IS during cognitive challenge.
Vaidya et al.23 reported that ADHD patients lacked right frontal lobe activation during RI and exhibited decreased activity of the left inferior frontal lobe during IS. Further, Bush et al.24 reported decreased cingulate cortex activity and atypical fronto-striatal activity during IS in adults with ADHD. Several studies25,26 have also reported that ADHD children exhibit relatively decreased fronto-striatal activity compared to other normal children. However, some studies have also reported increased activity or no difference.4 These controversial findings may stem from differences in the event-related paradigm. In a recent study, Bush et al.27 reported increases in the activity of the dorsal anterior midcingulate cortex, right dorsolateral prefrontal cortex, and both parietal lobes in adult ADHD patients on OROS MPH for 6 weeks. Also, during IS, ADHD patients are known to show atypical activation of the frontal lobe and caudate nucleus when compared to normal control group.23 In this study, altered activation of the caudate nucleus was not found; however, decreased activity of the frontal lobe was observed when MPH was discontinued as compared to when patients were on MPH. In particular, when subjects were on MPH, greater activation of the frontal lobe was observed during IS than when neutral stimulus was given (Figure 2). The authors could conclude that MPH has an effect on the right frontal lobe during IS test which corresponds with the results of Bush et al.27; however, changes in the right frontal lobe activity during an RI task with MPH administration were not seen in this study.
Scheres et al.5 reported that MPH had an affect on response time of RI rather than on IS and that this affect was unrelated to dose. On the other hand, in this study, only response time significantly different between on and off MPH during IS, a finding which is in contrast to previous studies. Further, although medication was discontinued for a week, considering inconsistent cognitive data with previous studies, the residual effect of long term medication treatment cannot be excluded. Finally, a dose-response relationship was not verified in this study.
In the current study, subjects exhibited symptomatic improvements with MPH; however, improved executive function of the frontal lobe was not observed except for the response time of IS. Further, MPH-induced increases in frontal lobe activity were identical to those in previous studies.23,28 Several studies10,29 have reported that the lateral frontal lobe may be the center for interference control in adults. This study replicated previous study results and suggested the possibility of concluding that MPH can normalize ADHD's cognitive control center. But unlike Bush et al.'s results,24,27 decreased activity in the anterior cingulate in adult with ADHD during IS was not observed.
The dopamine system affect on the fronto-sriatal network,12 as related to the RI model, can be activated by MPH treatment; however, this activated fronto-striatal network alone cannot fully explain improved IS. The role of the parieto-temporal network in IS improvement is also important.30,31 Unlike previous studies,5,32 the effect of MPH related to IS as opposed to RI was observed in this study.
Comprehensive results from the previous fMRI studies using event-related designs on cognitive controls25,30,33 have revealed the activation of widespread regions of the frontal cortex during RI. Particularly striking is the predominance of right-hemisphere activation, especially within the inferior frontal region. Recently, Bush et al.27 focused on changes in the right frontal lobe activity during IS in adults with ADHD on medication. This study also revealed increased right frontal lobe activity similar to that of healthy children during incongruent stimulus while being treated with MPH.
This study was limited in that it utilized a sample size that was too small to generalize the relationship between MPH and increased activity of the right frontal lobe. Further studies utilizing a larger sample size are needed. Moreover, there was no normal control group for head-to-head comparisons. A second limitation was that subjects were recruited from a population that had been undergoing treatment in clinical practice. That was why the second MRI scan was done one week after discontinuation of medication. However, it was impossible to completely exclude the possibility of a residual effect of longterm medication. Thus in further studies, drug-naïve patients should be selected for such studies and the first MRI should be performed prior to medication initiation and a subsequent MRI can proceed after the initiation of medication.
Acknowledgments
This research was financially supported by a grant (Choi, Sin-hae) from Korean Neuropsychiatry Association in 2006.
References
1. Barkley RA. Attention-deficit hyperactivity disorder: A clinical workbook. 1991,New York: Guilford Press.
2. Aron AR, Poldrack RA. The cognitive neuroscience of response inhibition: relevance for genetic research in attention-deficit/hyperactivity disorder. Biol Psychiatry 2005;57:1285-1292. PMID: 15950000.


3. Schachar R, Mota VL, Logan GD, Tannock R, Klim P. Confirmation of an inhibitory control deficit in attention-deficit/hyperactivity disorder. J Abnorm Child Psychol 2000;28:227-235. PMID: 10885681.


4. Vaidya CJ, Austin G, Kirkorian G, Ridlehuber HW, Desmond JE, Glover GH, et al. Selective effects of methylphenidate in attention deficit hyperactivity disorder: a functional magnetic resonance study. Proc Natl Acad Sci U S A 1998;95:14494-14499. PMID: 9826728.



5. Scheres A, Oosterlaan J, Swanson J, Morein-Zamir S, Meiran N, Schut H, et al. The effect of methylphenidate on three forms of response inhibition in boys with AD/HD. J Abnorm Child Psychol 2003;31:105-120. PMID: 12597703.


6. Konrad K, Neufang S, Fink GR, Herpertz-Dahlmann B. Long-term effects of methylphenidate on neural networks associated with executive attention in children with ADHD: results from a longitudinal functional MRI study. J Am Acad Child Adolesc Psychiatry 2007;46:1633-1641. PMID: 18030085.


7. Ridderinkhof KR, van der Molen MW, Band GP, Bashore TR. Sources of interference from irrelevant information: a developmental study. J Exp Child Psychol 1997;65:315-341. PMID: 9178963.


8. Huttenlocher PR, Dabholkar AS. Regional differences in synaptogenesis in human cerebral cortex. J Comp Neurol 1997;387:167-178. PMID: 9336221.


9. Minkowski A. Council for International Organizations of Medical S, France. D├®l├®gation g├®n├®rale ├Ā la recherche scientifique et t. Regional development of the brain in early life: a symposium organized by the Council for International Organizations of Medical Sciences. 1967,Oxford, Edinburgh: Blackwell Scientific.
10. Bunge SA, Dudukovic NM, Thomason ME, Vaidya CJ, Gabrieli JD. Immature frontal lobe contributions to cognitive control in children: evidence from fMRI. Neuron 2002;33:301-311. PMID: 11804576.



11. Luna B, Thulborn KR, Munoz DP, Merriam EP, Garver KE, Minshew NJ, et al. Maturation of widely distributed brain function subserves cognitive development. NeuroImage 2001;13:786-793. PMID: 11304075.


12. Castellanos FX, Giedd JN, Marsh WL, Hamburger SD, Vaituzis AC, Dickstein DP, et al. Quantitative brain magnetic resonance imaging in attention-deficit hyperactivity disorder. Arch Gen Psychiatry 1996;53:607-616. PMID: 8660127.


13. Castellanos FX, Giedd JN, Eckburg P, Marsh WL, Vaituzis AC, Kaysen D, et al. Quantitative morphology of the caudate nucleus in attention deficit hyperactivity disorder. Am J Psychiatry 1994;151:1791-1796. PMID: 7977887.


14. Dickstein SG, Bannon K, Castellanos FX, Milham MP. The neural correlates of attention deficit hyperactivity disorder: an ALE meta-analysis. J Child Psychol Psychiatry 2006;47:1051-1062. PMID: 17073984.


15. Rubia K, Smith AB, Brammer MJ, Toone B, Taylor E. Abnormal brain activation during inhibition and error detection in medication-naive adolescents with ADHD. Am J Psychiatry 2005;162:1067-1075. PMID: 15930054.


16. American Psychiatric Association. Task Force on D-I. Diagnostic and statistical manual of mental disorders: DSM-IV. 1994,Washington, DC: American Psychiatric Association.
17. Reid R, DuPaul GJ, Power TJ, Anastopoulos AD, Rogers-Adkinson D, Noll MB, et al. Assessing culturally different students for attention deficit hyperactivity disorder using behavior rating scales. J Abnorm Child Psychol 1998;26:187-198. PMID: 9650625.


18. Kim YS, Cheon KA, Kim BN, Chang SA, Yoo HJ, Kim JW, et al. The reliability and validity of Kiddie-Schedule for Affective Disorders and Schizophrenia-Present and Lifetime Version-Korean version (K-SADS-PL-K). Yonsei Med J 2004;45:81-89. PMID: 15004873.


19. Kwak K, Park H, Kim C. The manual for the Korean WISC-III. 2002,Seoul: The Special Education.
20. Shafritz KM, Marchione KE, Gore JC, Shaywitz SE, Shaywitz BA. The effects of methylphenidate on neural systems of attention in attention deficit hyperactivity disorder. Am J Psychiatry 2004;161:1990PMID: 15514398.


21. Zang YF, Jin Z, Weng XC, Zhang L, Zeng YW, Yang L, et al. Functional MRI in attention-deficit hyperactivity disorder: evidence for hypofrontality. Brain Dev 2005;27:544-550. PMID: 15876503.


22. Talairach J, Tournoux P. Co-planar stereotaxic atlas of the human brain: an approach to medical cerebral imaging. 1988,Stuttgart, New York, New York: G. Thieme, Thieme Medical Publishers.
23. Vaidya CJ, Bunge SA, Dudukovic NM, Zalecki CA, Elliott GR, Gabrieli JD. Altered neural substrates of cognitive control in childhood ADHD: evidence from functional magnetic resonance imaging. Am J Psychiatry 2005;162:1605-1613. PMID: 16135618.



24. Bush G, Frazier JA, Rauch SL, Seidman LJ, Whalen PJ, Jenike MA, et al. Anterior cingulate cortex dysfunction in attention-deficit/hyperactivity disorder revealed by fMRI and the Counting Stroop. Biol Psychiatry 1999;45:1542-1552. PMID: 10376114.


25. Durston S, Tottenham NT, Thomas KM, Davidson MC, Eigsti IM, Yang Y, et al. Differential patterns of striatal activation in young children with and without ADHD. Biol Psychiatry 2003;53:871-878. PMID: 12742674.


26. Rubia K, Overmeyer S, Taylor E, Brammer M, Williams SC, Simmons A, et al. Hypofrontality in attention deficit hyperactivity disorder during higher-order motor control: a study with functional MRI. Am J Psychiatry 1999;156:891PMID: 10360128.


27. Bush G, Spencer TJ, Holmes J, Shin LM, Valera EM, Seidman LJ, et al. Functional magnetic resonance imaging of methylphenidate and placebo in attention-deficit/hyperactivity disorder during the multisource interference task. Arch Gen Psychiatry 2008;65:102-114. PMID: 18180434.


28. Lijffijt M, Kenemans JL, ter Wal A, Quik EH, Kemner C, Westenberg H, et al. Dose-related effect of methylphenidate on stopping and changing in children with attention-deficit/hyperactivity disorder. Eur Psychiatry 2006;21:544-547. PMID: 15994064.


29. Peterson BS, Potenza MN, Wang Z, Zhu H, Martin A, Marsh R, et al. An FMRI study of the effects of psychostimulants on default-mode processing during Stroop task performance in youths with ADHD. Am J Psychiatry 2009;166:1286-1294. PMID: 19755575.



30. Bunge SA, Hazeltine E, Scanlon MD, Rosen AC, Gabrieli JD. Dissociable contributions of prefrontal and parietal cortices to response selection. Neuroimage 2002;17:1562-1571. PMID: 12414294.


31. Casey BJ, Thomas KM, Welsh TF, Badgaiyan RD, Eccard CH, Jennings JR, et al. Dissociation of response conflict, attentional selection, and expectancy with functional magnetic resonance imaging. Proc Natl Acad Sci U S A 2000;97:8728-8733. PMID: 10900023.



32. Aron AR, Dowson JH, Sahakian BJ, Robbins TW. Methylphenidate improves response inhibition in adults with attention-deficit/hyperactivity disorder. Biol Psychiatry 2003;54:1465-1468. PMID: 14675812.


33. Rubia K, Smith AB, Brammer MJ, Taylor E. Right inferior prefrontal cortex mediates response inhibition while mesial prefrontal cortex is responsible for error detection. NeuroImage 2003;20:351-358. PMID: 14527595.


Figure┬Ā1
Example of a modified Eriksen flanker task. Each trial consisted of a display comprising a central arrow and two flanking stimuli on either side. Subjects were instructed to press one of two buttons indicating the direction of the central arrow (left/right) while ignoring the flanking stimuli. On congruent and incongruent trials, the flankers were arrows pointing in the same or different direction respectively in relation to the central arrow. On neutral trials the flankers were diamonds that were not associated with a response. On no-go trials, the flanking stimuli signaled were Xs that subjects should withhold their response.

Figure┬Ā2
Neural correlates of interference suppression during incongruent relative to neutral trials between children with ADHD on MPH treatment. Images show region of more activation during incongruent relative to neutral trials on MPH treatment. For regions positively correlated with improved interference suppression, uncorrected p<0.005. ADHD: attention deficit hyperactivity disorder, MPH: methylphenidate.