|
 |
- Search
| Psychiatry Investig > Volume 9(3); 2012 > Article |
Abstract
Objective
The primary aim of this study was to compare electronic monitoring with other measures of adherence to Osmotic-controlled Release Oral delivery System methylphenidate in children with attention-deficit hyperactivity disorder (ADHD). The secondary aim was to analyze the relationships between adherence and clinical factors, including ADHD symptoms.
Methods
Thirty-nine children diagnosed with ADHD were monitored for adherence to medication over the course of eight weeks. Medication adherence was assessed using the Medication Event Monitoring System (MEMS), which is a bottle cap with a microprocessor that records all instances and times that the bottle is opened; patient self-report; clinician rating; and pill count. Information, including demographic and clinical characteristics, symptom rating scale, and psychological test results, were also collected. The relationships between adherence and clinical factors, including ADHD rating scores of baseline and of the changes, were assessed.
Results
The rate of non-adherence measured by the MEMS was found to be 46.2%, which was considerably higher than those of the patient self-report (17.9%), clinician rating (31.7%), and pill count (12.8%) of non-adherence. The rate of adherence measured by the MEMS was not significantly associated with baseline symptom severity or symptom changes over the eight weeks, although non-adherent group showed more severe baseline symptoms and inferior improvement.
Conclusion
Adherence as measured by the MEMS showed a discrepancy with other measures of adherence in patients with ADHD. The symptom severity and level of improvement were not related to adherence with MEMS. Further studies are needed to evaluate the variables that may impact medication adherence in children with ADHD.
It is estimated that approximately 5-10% of school-aged children suffer from attention-deficit hyperactivity disorder (ADHD).1,2 If left untreated, ADHD can have a debilitating impact on normal development, with affected children more likely to have issues with school performance and peer and family relationships,1,2 even persisting throughout adolescence and adulthood. Considering the chronic nature of ADHD and the serious functional limitations and impairments linked to untreated ADHD, poor adherence to medication in children with ADHD may be a significant barrier to positive treatment outcome.3 Not only will children with untreated or poorly treated ADHD potentially experience continued issues in many areas of their life, but health care providers may also experience difficulties in determining treatment efficacy and assessing the need for medication changes.4
Adherence is defined as the extent to which a patient's actions correspond to the treatment recommendations of health care providers.5 Rates of stimulant adherence in children with ADHD vary considerably across samples and studies,6-11 most likely owing to differences in the study period and in defining and measuring adherence.3,4 Data from community-based samples indicate that the average time to medication discontinuation is four months,10 and that families are fully adherent with the treatment regimen for an average of only two months.12 Faraone et al.8 found that up to 86% of subjects continued medication at the end of one year. More typically, however, adherence rates in clinical research samples are approximately 50-80% after one year and 36-46% after five years.6,13
Assessment methods used to examine medication adherence in pediatric populations have included child report, parent report, pill count, and measurement of medication concentrations in urine or blood samples. Overestimation of adherence is common when using direct reports from parents or children or when using pill counts.14 Measuring concentrations of medication in urine or blood samples is a resource-consuming method, and collecting samples can be invasive and uncomfortable for the children. The Medication Event Monitoring System (MEMS; Aardex Ltd., Union City, CA, USA) is a device that records the date and time the pill container was opened using an electronic computer chip implanted into the cap of a prescription bottle. Data are downloaded directly into a computer program.15 This tool reliably assesses medication use in clinical and research settings and is regarded as the gold standard for evaluating adherence.16 In this study, this electronic device was used to evaluate stimulant adherence in children with ADHD.
Studies on adherence in patients with ADHD suggest that predictors of poor adherence may include older age,3,13 male gender,17 lower intelligence,14,17 oppositional-defiant symptoms,13 lower socioeconomic status,9 and a three-times-daily regimen. Additionally, patients who experience more severe symptoms are more likely to adhere to their medication,13 while patients who experience fewer symptoms exhibit lower adherence.8 Those who are adherent to their stimulant treatment are more likely to have higher symptom scores over long-term follow-up than are non-adherents.6 This relation between symptom severity and adherence suggests that a significant improvement in symptoms in response to medication likely motivates families to continue to use medication. The primary aim of this prospective study was to examine differences in adherence to Osmotic-controlled Release Oral delivery System (OROS) methylphenidate as estimated by self-report, a clinician's rating scale, pill count, and MEMS in children with ADHD. The secondary aim was to explore the relationships between adherence and clinical factors including ADHD subtype, onset and duration of illness, symptom severity, and neuropsychological test results.
Subjects were recruited consecutively from outpatient clinics at the University Hospital in Seoul, Korea. Diagnostic interviews of children with ADHD and one or both parents were conducted by child-adolescent psychiatrists. Inclusion criteria were as follows: 1) diagnosis of ADHD based on the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV), 2) capacity to understand and communicate with investigators, 3) age ranging from 7-13 years, and 4) current use of an OROS methylphenidate on fixed dose of medication, not during the titration period. Patients with co-morbid diagnoses that may have influenced outcome measures (e.g., mental retardation or pervasive developmental disorder), severe medical conditions, or seizure disorder were excluded; however, patients with mild symptoms of depression or tics were included. Only children with ADHD who did not require dose adjustment for the eight weeks of the study were included. Patients who failed to attend study visits were excluded from the analysis. Appointments that were early or delayed by two or three days were allowed. Informed consent was provided both by the children and their parents. The study was approved by the Research and Ethics Committee of Korea University.
The following variables were collected via clinician interview and chart review at baseline to test for potential associations with prospectively measured adherence: age, gender, height, weight, parental education, marital status of parents, and income. Information regarding the subtype of ADHD based on DSM-IV criteria, age of onset, dosage of OROS methylphenidate, and length of treatment with medication were also obtained. The total number of tablets consumed was determined at each visit.
Parental versions of the ADHD Rating Scale (ARS) were used in order to evaluate the symptoms of children with ADHD. Assessments were carried out at baseline and at the end of the eight week study period.
Adherence was assessed using the following four methods: MEMS, patient self-report, a clinician rating scale, and pill count.
The percentage of doses taken on schedule was used to calculate the MEMS adherence rate. Subjects were categorized as adherent if they opened the pill container within the 3-hour target time frame for each dose.
Percentage of doses taken on schedule (PDTc, %)=No. of doses taken correctly according to the prescription/No. of prescribed doses×100
Parents of the children with ADHD were asked to estimate their child's overall adherence to the stimulant medication with a value between 0 and 100%.
The clinician rating scale was an ordinal scale from 1-7, with higher numbers indicating better adherence (response range: 1 'complete refusal' to 7 'active participation: readily accepts, and shows some responsibility for regimen'). In previous studies, a score of 5 or greater was used to determine adherence,20 and this study adopted the same threshold for adherence. Clinician assessment of adherence was conducted without knowledge of the MEMS data.
Adherence was analyzed in two ways. First, adherence was treated as a continuous variable. Second, the continuous adherence data were converted into dichotomous variables, with the exception of the clinician rating scale. A threshold of 80% was used, which is considered the standard cut-off for a dichotomous measurement of adherence.15,21 Subjects were categorized as adherent and non-adherent based on MEMS adherence.22 Chi-square tests and independent t-tests were used to compare the demographic and clinical variables of adherent and non-adherent groups, respectively. Pearson's correlation was used to determine the association between adherence rates as defined by the MEMS and demographic and clinical factors. Mantel-Haenszel tests of linear association were also used to estimate the relationships between adherence and ordinal variables. A concordance correlation analysis was used to evaluate agreement among the adherence measures. The concordance correlation coefficient assesses the agreement between two methods by measuring the variation in their linear relationship from the 45° line through the origin.23 Kappa statistics were calculated to evaluate agreement among the dichotomized adherence measures. p-values less than 0.05 according to a two-tailed test were considered statistically significant. Data were analyzed using the Statistical Packages for the Social Sciences (SPSS version 16; SPSS Inc., Chicago, IL, USA).
A total of 44 outpatients (39 boys, 5 girls) were enrolled in the study. Two did not return the MEMS bottle at the follow-up visit, and two failed to attend their appointment on the scheduled day. One other did not use the MEMS bottle consistently and returned with unusable data. The final sample consisted of 39 children (35 boys, 4 girls) with a mean age of 10.44±2.22 years. Mean height was 138.21±14.50 cm, and mean weight was 36.76±15.17 kg. Mean dose of OROS methylphenidate was 33.23±13.25 mg. Concomitant medications that were taken during the study included escitalopram (n=5), risperidone (n=2), and imipramine (n=1). There were no differences in baseline demographic or clinical characteristics between the adherent and non-adherent groups (Table 1).
Mean adherence according to the MEMS, self-report, and pill count was 77.58±19.13%, 90.08±16.25%, and 94.06±8.62%, respectively. The mean scale score for the clinician rating scale of adherence was 4.58±0.99. By employing a score of 5 or greater as the threshold for clinically meaningful adherence, the clinician rating scale of adherence was low. Concordance correlations (95% confidence interval) among the adherence variables were low: 0.237 (range of -0.024 to 0.468) for self-report vs. MEMS, 0.249 (0.102 to 0.386) for pill count vs. MEMS, and 0.269 (0.009 to 0.494) for self-report vs. pill count.
Continuous adherence data were converted into dichotomous variables (adherent vs. non-adherent group). Employing this approach, the adherence rates for the MEMS, clinician rating scale, self-report, and pill count were 53.8%, 68.3%, 82.1%, and 87.2%, respectively. Kappa coefficients indicated that agreement among adherence measures was low: 0.083 for self-report and MEMS, 0.293 for pill count and MEMS, and 0.327 for clinician rating scale and MEMS.
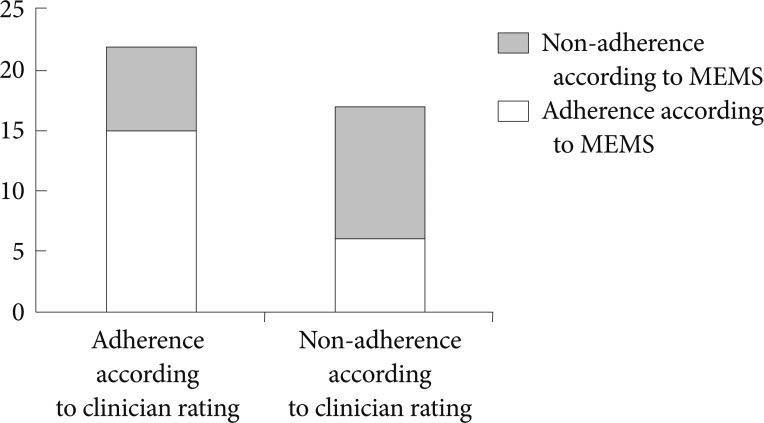
Seven of the 22 subjects who were rated as adherent by their treating clinician were categorized as non-adherent based on the MEMS, and six of the 17 who were rated as non-adherent by their treating clinician were categorized as adherent based on the MEMS (Figure 1). Thirty-three percent (13/39) of the clinician adherence ratings did not agree with adherence as determined by the MEMS.
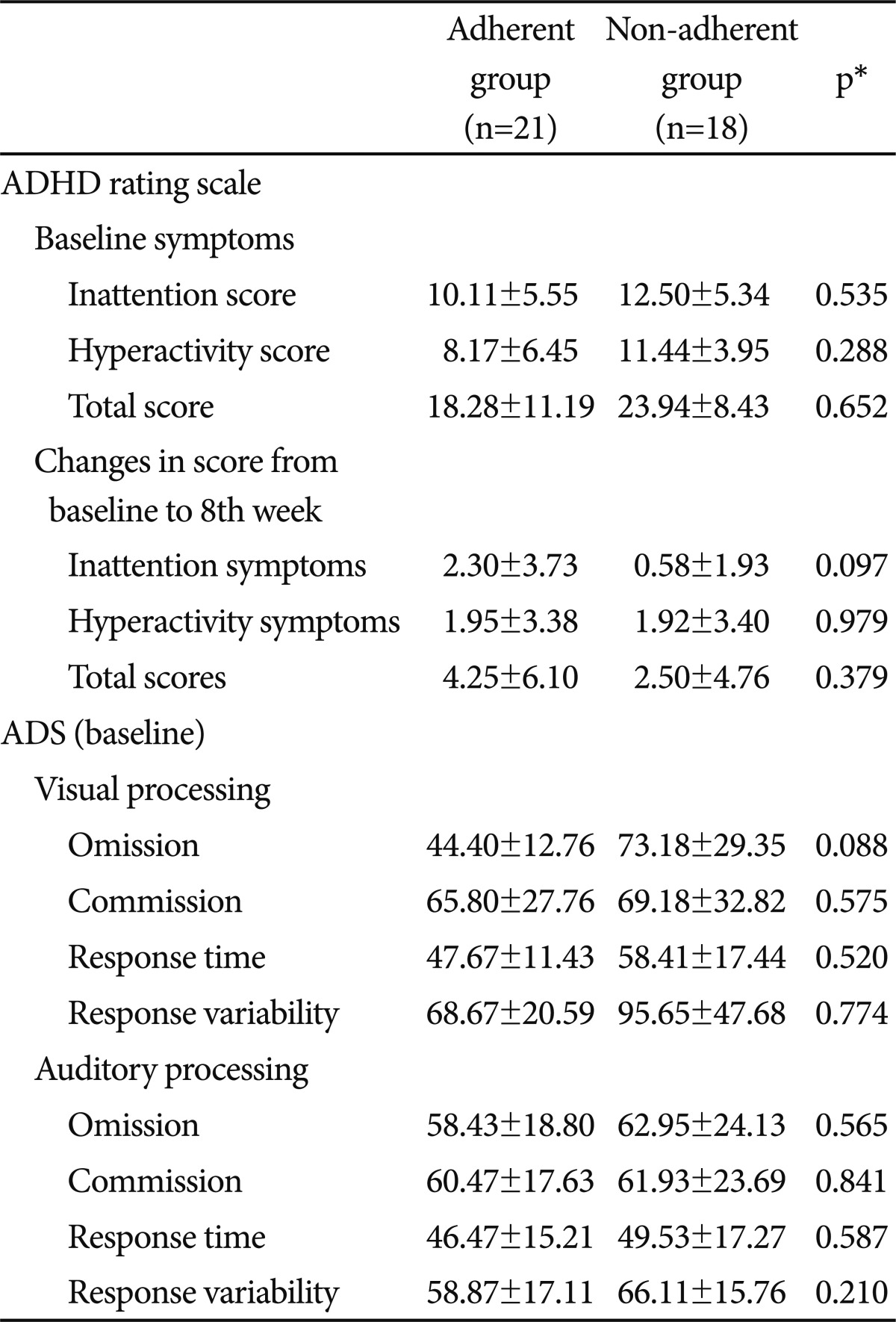
The relationships between adherence variables and demographic and clinical factors were examined. Baseline ARS scores and ARS score changes were compared between the adherent and non-adherent groups as defined by the MEMS (Table 2). There were no significant differences between the two groups, although the non-adherent group reported higher scores both at baseline scores and score changes on the ARS. All ARS subscale scores decreased in both groups from baseline to the eight-week follow-up (p<0.05); however, these changes did not differ between the two groups even though the adherent group showed a larger mean decrease in score.
In the correlation analysis, the inattention subscale and total scores of the ARS at baseline were negatively correlated with self-rated adherence (ARS inattention: r2=-0.403, p<0.05; ARS total: r2=-0.385, p<0.05). However, none of these measures were significantly correlated with MEMS adherence.
Baseline intelligence test scores and CPT scores were not different between adherent and non-adherent groups. Moreover, there were no correlations between MEMS adherence and these scores.
In the present study, the MEMS monitoring detected clinically meaningful OROS methylphenidate non-adherence in nearly 50% of the subjects when adherence was defined as a dichotomous variable. Using a threshold of 80%, the rate of non-adherence as measured by the MEMS was 46.2%, which was considerably higher than non-adherence based on self-report (17.9%), clinician ratings (31.7%), and pill count (12.8%). Inconsistency between self-report and clinician estimates of adherence has been previously reported;24 however, the present study suggests that the ability of clinicians as well as patients to predict adherence is limited. In addition to an overestimation of patient adherence, there was a discrepancy between adherence measures according to clinician rating and the MEMS, which is a more accurate measure of adherence; One-third of clinician adherence ratings did not agree with adherence as determined by the MEMS. Moreover, the clinician rating scores had a low kappa coefficient with MEMS in the correlation analysis. These findings suggest the possibility of a limited clinician capacity to detect OROS methylphenidate non-adherence in actual clinical practice. Also, the clinicians needed additional time in the follow-up interview for determining non-adherence.
Moreover, pill count adherence was even higher than self-reported adherence, and there was a significant discrepancy in adherence calculated by pill count and adherence calculated with the MEMS. In this study, the MEMS considered a patient adherent when the bottle was opened within a specified 3-hour time frame. The discrepancy between pill count and MEMS adherence implies that the medications were not taken within the recommended time frame. Dosing time may be particularly important in children with ADHD. Delayed dosing can result in insufficient treatment effects or adverse effects. Parents of children with ADHD often consider their child adherent even though the child took the medication at the incorrect time. The MEMS provides more details than other measures of adherence and accounts for intake pattern.23 The discrepancies in the results of the present study highlight the importance of the criteria used to define adherence. The utility of the MEMS is its potential for measuring patterns of adherence and its ability to encourage adherence in children and their parents, since symptom control and adverse effects of stimulants are more closely linked to taking medication on time, as opposed to a pattern of missed doses. Moreover, there may be subgroups of individuals who respond differently to changes in medication regime, ranging from those who are sensitive to even the slightest changes to those who maintain a response despite frequent non-adherence.25 The MEMS reliably measures medication use in clinical and research settings, and comparisons of MEMS adherence with other measures can be meaningful for educating and improving adherence in clinical practice.
In the current study, ADHD symptoms and their changes with respect to adherence were analyzed. The relationships between adherence and ADHD symptoms in the children with ADHD were also evaluated. An analysis showed that adherence as defined by the MEMS did not have a significant relationship with baseline symptom severity or symptom changes during the eight week study. That said, inattention and the total symptom scores that patients experienced were negatively associated with self-reported adherence. This suggests that patients as well as clinicians may assume that adherence is related to symptoms. That is, patients may think that symptom improvement is due to good adherence or that good adherence will result in improvement of their symptoms in the short-term.
In the study of the relationship between adherence and symptoms, some have suggested that baseline severity of ADHD predicts adherence to medication treatment. Brown et al. found that more severe symptoms were associated with lower adherence rates.14 Another study found that a greater number of teacher-rated ADHD symptoms at baseline predicted adherence to therapy.13 More recently, Faraone et al. reported that fewer ADHD symptoms were associated with low adherence.8 The differences in the definition and measure of adherence may be due to the source of these contradictory results. In the present study, there were no significant differences in baseline symptom scores or symptom change scores with ARS between the adherent and non-adherent groups; however, the non-adherent group showed higher baseline scores and smaller changes on the ARS during the eight week study period. We tentatively suggest that the severe baseline symptoms and poor improvement may be related to non-adherence.
The results of this study should be interpreted in light of several study limitations. First, although the MEMS is currently considered the gold standard for evaluating adherence, it has limitations. Documentation that the bottle was opened does not confirm that either the medication or the correct dose was ingested. Secondly, although subjects were not informed of the results of the MEMS measurements, they were aware that this study's intent was to better understand adherence, which may have encouraged adherence during the study. Thirdly, subjects with a co-morbid disorder such as mild symptoms of depression or tics were included, and these comorbid cases many influence the adherence to medication. Fourth, the relatively small number of participants also may have influenced the study results.
This is the first study to examine adherence using the MEMS in addition to clinician ratings and pill count measures in children with ADHD. This study is also unique in that it assessed the relationship between adherence estimated with MEMS and symptoms in children with ADHD. Based on the MEMS data, the overall mean level of adherence was low, suggesting that clinician- or parent-ratings and pill counts may overestimate adherence. The symptom severity and level of improvement were not related to adherence estimated with MEMS. More studies are needed to evaluate other variables that may predict treatment adherence in ADHD. Increased use of more precise evaluation tools, such as the MEMS, may help resolve this matter.
References
1. Faraone SV, Sergeant J, Gillberg C, Biederman J. The worldwide prevalence of ADHD: is it an American condition? World Psychiatry 2003;2:104-113. PMID: 16946911.
2. Ahn DH, Kang H, Kim BN, Kim JH, Shin D, Yang SJ, et al. The Korean Practice Parameter for the treatment of attention-deficit hyperactivity disorder (I)-introduction, clinical features and course. J Korean Acad Child Adolesc Psychiatry 2007;18:3-9.
3. Gau SS, Shen HY, Chou MC, Tang CS, Chiu YN, Gau CS. Determinants of adherence to methylphenidate and the impact of poor adherence on maternal and family measures. J Child Adolesc Psychopharmacol 2006;16:286-297. PMID: 16768636.


4. Adler LD, Nierenberg AA. Review of medication adherence in children and adults with ADHD. Postgrad Med 2010;122:184-191. PMID: 20107302.


6. Charach A, Ickowicz A, Schachar R. Stimulant treatment over five years: adherence, effectiveness, and adverse effects. J Am Acad Child Adolesc Psychiatry 2004;43:559-567. PMID: 15100562.


7. Chou WJ, Chou MC, Tzang RF, Hsu YC, Gau SS, Chen SJ, et al. Better efficacy for the osmotic release oral system methylphenidate among poor adherents to immediate-release methylphenidate in the three ADHD subtypes. Psychiatry Clin Neurosci 2009;63:167-175. PMID: 19335386.


8. Faraone SV, Biederman J, Zimmerman B. An analysis of patient adherence to treatment during a 1-year, open-label study of OROS methylphenidate in children with ADHD. J Atten Disord 2007;11:157-166. PMID: 17494833.


9. Ibrahim el SR. Rates of adherence to pharmacological treatment among children and adolescents with attention deficit hyperactivity disorder. Hum Psychopharmacol 2002;17:225-231. PMID: 12404679.


10. Marcus SC, Wan GJ, Kemner JE, Olfson M. Continuity of methylphenidate treatment for attention-deficit/hyperactivity disorder. Arch Pediatr Adolesc Med 2005;159:572-578. PMID: 15939858.


11. Pappadopulos E, Jensen PS, Chait AR, Arnold LE, Swanson JM, Greenhill LL, et al. Medication adherence in the MTA: saliva methylphenidate samples versus parent report and mediating effect of concomitant behavioral treatment. J Am Acad Child Adolesc Psychiatry 2009;48:501-510. PMID: 19307987.


12. Perwien A, Hall J, Swensen A, Swindle R. Stimulant treatment patterns and compliance in children and adults with newly treated attention-deficit/hyperactivity disorder. J Manag Care Pharm 2004;10:122-129. PMID: 15032561.


13. Thiruchelvam D, Charach A, Schachar RJ. Moderators and mediators of long-term adherence to stimulant treatment in children with ADHD. J Am Acad Child Adolesc Psychiatry 2001;40:922-928. PMID: 11501692.


14. Brown RT, Borden KA, Clingerman SR. Adherence to methylphenidate therapy in a pediatric population: a preliminary investigation. Psychopharmacol Bull 1985;21:28-36. PMID: 3983335.

15. Charach A, Gajaria A, Skyba A, Chen S. Documenting adherence to psychostimulants in children with ADHD. J Can Acad Child Adolesc Psychiatry 2008;17:131-136. PMID: 18769643.


16. Claxton AJ, Cramer J, Pierce C. A systematic review of the associations between dose regimens and medication compliance. Clin Ther 2001;23:1296-1310. PMID: 11558866.


17. Firestone P. Factors associated with children's adherence to stimulant medication. Am J Orthopsychiatry 1982;52:447-457. PMID: 7114173.



18. Park KS, Yoon JY, Park HJ, Park HJ, Kwon KW. Korean Educational Development Institute-Wechsler Intelligence Scale for Children. 1991,Seoul: Korean Educational Development Institute.
19. Shin MS, Cho SZ, Chun SY, Hong KE. A study of the development and standardization of ADHD diagnositc system. J Child Adolesc Psychiatry 2000;11:91-99.
20. Byerly M, Fisher R, Whatley K, Holland R, Varghese F, Carmody T, et al. A comparison of electronic monitoring vs. clinician rating of antipsychotic adherence in outpatients with schizophrenia. Psychiatry Res 2005;133:129-133. PMID: 15740989.


21. Charach A, Gajaria A. Improving psychostimulant adherence in children with ADHD. Expert Rev Neurother 2008;8:1563-1571. PMID: 18928348.


22. Haynes R, Taylor D, Sackett D. Compliance in Health Care. 1979,Baltimore: Johns Hopkins University Press.
23. Lin LI. A concordance correlation coefficient to evaluate reproducibility. Biometrics 1989;45:255-268. PMID: 2720055.


24. Valenstein M, Barry KL, Blow FC, Copeland L, Ullman E. Agreement between seriously mentally ill veterans and their clinicians about medication compliance. Psychiatr Serv 1998;49:1043-1048. PMID: 9712210.


25. Remington G, Kwon J, Collins A, Laporte D, Mann S, Christensen B. The use of electronic monitoring (MEMS) to evaluate antipsychotic compliance in outpatients with schizophrenia. Schizophr Res 2007;90:229-237. PMID: 17208414.