 |
 |
- Search
| Psychiatry Investig > Volume 20(8); 2023 > Article |
|
Abstract
Objective
Methods
Notes
Availability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: Shunhua Zhang. Data curation: Tian Qin, Shunhua Zhang. Formal analysis: all authors. Funding acquisition: Shunhua Zhang. Investigation: all authors. Methodology: Tian Qin, Shunhua Zhang. Project administration: Shunhua Zhang. Resources: Shunhua Zhang. Software: all authors. Supervision: Shunhua Zhang. Validation: Tian Qin, Shunhua Zhang. Visualization: Tian Qin, Shunhua Zhang. WritingŌĆöoriginal draff: Tian Qin. WritingŌĆöreview & editing: Shunhua Zhang.
Funding Statement
This work was funded by the National Innovation and Entrepreneurship Training Program for college students of China under Grant 201910367009, 202010367036, 202110367049, and 202210367022, as well as Anhui Provincial Colleges and Universities Ideological and Political Work Ability Improvement Project (No. sztsjh2019-7-14), and the Key Projects of Teaching Research for Colleges and Universities in Anhui Province, China (No. 2020jyxm1174).
ACKNOWLEDGEMENTS
Figure┬Ā1.
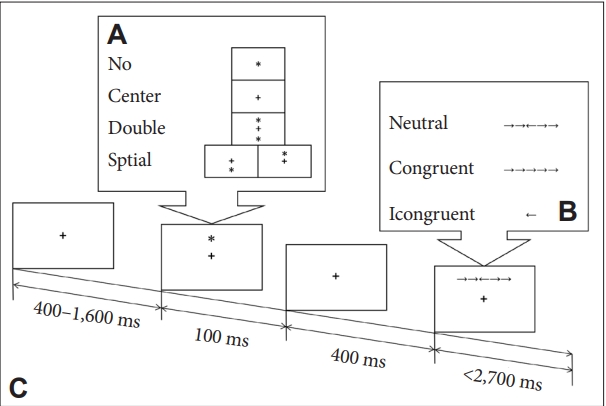
Figure┬Ā2.

Table┬Ā1.
| Measure | ASD parents (N=28) | TDC parents (N=28) | t or Žć2 | p | ||
|---|---|---|---|---|---|---|
| Demographic data | ||||||
| Age (yr) | 34.43┬▒6.14 | 34.11┬▒3.88 | 0.234* | 0.816 | ||
| Gender, man/woman | 9/19 | 8/20 | 0.084ŌĆĀ | 0.771 | ||
| Education (yr) | 12.36┬▒2.95 | 13.21┬▒3.46 | -0.998* | 0.323 | ||
| Neuropsychological performances | ||||||
| DS-Forward | 8.04┬▒0.10 | 8.42┬▒1.14 | -1.374* | 0.175 | ||
| DS-Backward | 5.50┬▒0.92 | 5.89┬▒1.40 | -1.242* | 0.220 | ||
| TMT-A (sec) | 44.22┬▒12.82 | 39.77┬▒14.35 | 1.227* | 0.225 | ||
| TMT-B (sec) | 85.13┬▒25.14 | 74.86┬▒18.03 | 1.758* | 0.084 | ||
| MoCA | 26.54┬▒1.90 | 27.00┬▒1.36 | -1.053* | 0.297 | ||
| Total AQ | 111.98┬▒12.15 | 110.61┬▒9.55 | 0.469* | 0.641 | ||
| Social skill | 22.93┬▒4.58 | 21.71┬▒3.81 | 1.079* | 0.285 | ||
| Attention switching | 25.07┬▒2.65 | 24.71┬▒3.48 | 0.432* | 0.668 | ||
| Attention to detail | 22.44┬▒4.74 | 23.00┬▒3.92 | -0.483* | 0.631 | ||
| Communication | 20.85┬▒4.86 | 20.36┬▒4.53 | 0.398* | 0.692 | ||
| Imagination | 20.68┬▒2.38 | 20.82┬▒2.93 | -0.195* | 0.846 | ||
There are no statistical differences between the two groups in demographic data and neuropsychological performances (all p>0.05).
ASD, autism spectrum disorder; TDC, typical developmental children; DS, The Digital Span Test; TMT, Trail Making Test; MoCA, Montreal Cognitive Assessment; AQ, Autism Spectrum Quotient
Table┬Ā2.
| ASD parents | TDC parents | t | p | |
|---|---|---|---|---|
| Alerting (ms) | 26.00┬▒22.31 | 41.79┬▒19.77 | 2.80 | 0.007** |
| Orienting (ms) | 36.39┬▒16.46 | 34.54┬▒24.37 | -0.33 | 0.740 |
| Executive control (ms) | 121.79┬▒32.84 | 98.89┬▒31.97 | -2.64 | 0.011* |
| Overall mean RT (ms) | 646.21┬▒90.40 | 653.25┬▒73.59 | 1.03 | 0.751 |
| Accuracy (%) | 98.82┬▒1.28 | 99.11┬▒0.74 | 0.32 | 0.310 |
Table┬Ā3.
|
ASD parents |
TDC parents |
|||||
|---|---|---|---|---|---|---|
| Alerting | Orienting | Executive | Alerting | Orienting | Executive | |
| Alerting | 1 | 1 | ||||
| Orienting | 0.274 | 1 | 0.148 | 1 | ||
| Executive | 0.179 | 0.005 | 1 | -0.001 | -0.089 | 1 |
Table┬Ā4.
There are no statistical correlations between the three subtypes of attention network and various dimensions of AQ for both groups (p>0.05). ASD, autism spectrum disorder; TDC, typical developmental children; AQ, Autism Spectrum Quotient; AQ-total, total AQ; AQ-S, AQ-social skill; AQ-AS, AQ-attention switch; AQ-AD, AQ-attention to detail; AQ-C, AQ-communication; AQ-IM, AQ-imagination
REFERENCES
-
METRICS

-
- 0 Crossref
- Scopus
- 1,810 View
- 38 Download
-
Exposure to Environmental Toxins in Mothers of Children with Autism Spectrum Disorder2010 June;7(2)






