 |
 |
- Search
| Psychiatry Investig > Volume 19(10); 2022 > Article |
|
Abstract
Objective
Methods
Results
Notes
Availability of Data and Material
Data used in this study are available at the AlzheimerŌĆÖs Disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu).
Conflicts of Interest
The authors have no potential conflicts of interest to disclose.
Author Contributions
Conceptualization: all authors. Data curation: Young Min Choe. Formal analysis: Young Min Choe, Jee Wook Kim. Investigation: Young Min Choe, Guk-Hee Suh. Methodology: Young Min Choe. Software: Young Min Choe. Supervision: Jee Wook Kim. Validation: Guk-Hee Suh, Jee Wook Kim. WritingŌĆöoriginal draf: Young Min Choe. WritingŌĆöreview & editing: Guk-Hee Suh, Jee Wook Kim.
Funding Statement
This study was supported by Hallym University Research Fund (HURF2020-34). Data collection and sharing for this project was funded by the AlzheimerŌĆÖs Disease Neuroimaging Initiative (ADNI) (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, AlzheimerŌĆÖs Association; AlzheimerŌĆÖs Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (www.fnih.org). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the AlzheimerŌĆÖs Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
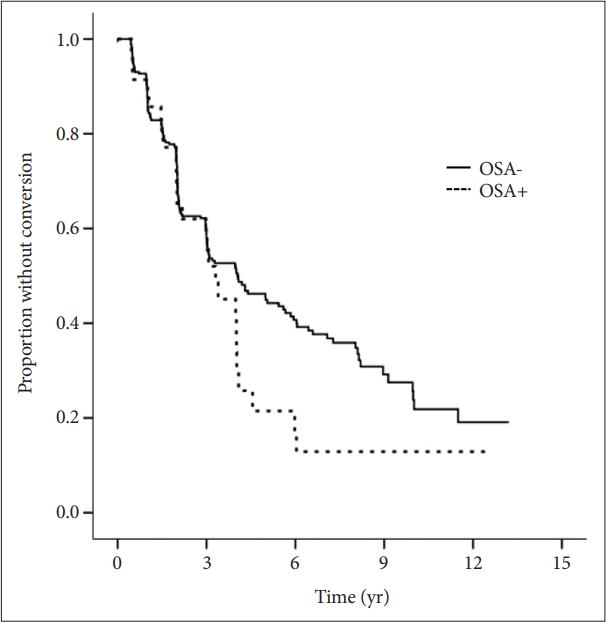
Figure┬Ā1.
Table┬Ā1.
| All (N=1,058) | CN (N=391) | EMCI (N=303) | LMCI (N=364) | p | |
|---|---|---|---|---|---|
| Age (yr) | 73.03┬▒6.96 | 73.72┬▒5.59 | 71.28┬▒7.37 | 73.74┬▒7.41 | <0.001*ac |
| Sex, male | 587 (55.5) | 195 (49.9) | 169 (55.8) | 223 (61.3) | 0.007* |
| Education level (yr) | 16.19┬▒2.72 | 16.43┬▒2.72 | 15.97┬▒2.65 | 16.10┬▒2.77 | 0.504 |
| Apolipoprotein E ╬Ą4 carrier | 445 (42.1) | 113 (28.9) | 131 (43.2) | 201 (55.2) | <0.001* |
| Vascular risk score | 1.01┬▒0.98 | 0.97┬▒0.95 | 1.05┬▒0.99 | 1.03┬▒1.00 | 0.100 |
| Body mass index (kg/m2) | 27.28┬▒4.98 | 27.22┬▒5.16 | 28.01┬▒5.32 | 26.74┬▒4.40 | 0.005*c |
| MMSE | 28.24┬▒1.67 | 29.00┬▒1.21 | 28.34┬▒1.57 | 27.35┬▒1.77 | <0.001*abc |
| Geriatric Depression Scale | 1.39┬▒1.41 | 0.87┬▒1.16 | 1.77┬▒1.51 | 1.65┬▒1.40 | <0.001*ab |
| Clinical conversion | 326 (30.8) | 86 (22.0) | 52 (17.2) | 188 (51.6) | <0.001* |
| Sleep disorder | 178 (16.8) | 48 (12.3) | 74 (24.4) | 56 (15.4) | <0.001* |
| ŌĆāInsomnia | 76 (7.2) | 22 (5.6) | 33 (10.9) | 21 (5.8) | 0.013* |
| ŌĆāOSA | 101 (9.5) | 22 (5.6) | 44 (14.5) | 35 (9.6) | <0.001* |
| ŌĆāRLS | 16 (1.5) | 7 (1.8) | 5 (1.7) | 4 (1.1) | 0.719 |
| Use of sleeping pill | 88 (8.3) | 36 (9.2) | 33 (10.9) | 19 (5.2) | 0.022* |
Table┬Ā2.
| B | SE | Wald | OR | 95% CI | p | |
|---|---|---|---|---|---|---|
| Sleep disorder | 0.671 | 0.204 | 10.826 | 1.956 | 1.312-2.917 | <0.001* |
| ŌĆāInsomnia | 0.647 | 0.300 | 4.657 | 1.910 | 1.061-3.436 | 0.031* |
| ŌĆāOSA | 0.775 | 0.268 | 8.396 | 2.172 | 1.285-3.669 | 0.004* |
| ŌĆāRLS | -0.067 | 0.548 | 0.015 | 0.935 | 0.320-2.735 | 0.903 |
Table┬Ā3.
|
Model 1 |
Model 2 |
|||
|---|---|---|---|---|
| OR (95% CI) | p | OR (95% CI) | p | |
| Sleep disorder | 1.194 (0.820-1.739) | 0.356 | 1.409 (0.945-2.099) | 0.092 |
| ŌĆāInsomnia | 0.585 (0.316-1.084) | 0.089 | 0.605 (0.314-1.163) | 0.132 |
| ŌĆāOSA | 1.772 (1.113-2.820) | 0.016* | 2.073 (1.282-3.354) | 0.003* |
| ŌĆāRLS | 1.221 (0.399-3.738) | 0.727 | 1.522 (0.491-4.716) | 0.467 |
Table┬Ā4.
Adjusted for age, sex, education, apolipoprotein E ╬Ą4 status, vascular risk score, body mass index, geriatric depression scale, use of sleeping pill, and baseline Mini-Mental State Examination score. *p<0.05. OSA, obstructive sleep apnea; RLS, restless legs syndrome; CN, cognitively normal; OR, odds ratio; CI, confidence interval; EMCI, early mild cognitive impairment; LMCI, late mild cognitive impairment
REFERENCES







